Advertisement|Remove ads.
ATRA Stock Sees Biggest Single-Day Gain In Over 2 Years, Breaches Key Technical Milestone – New FDA Update Has Bulls Circling Back

- Atara is jointly developing the drug with Pierre Fabre Pharmaceuticals.
- The FDA stated that a single-arm study using a historical control group could support a future marketing application.
- FDA had rejected an initial application submitted by Atara in January 2026, raising concerns about the trial design and analysis.
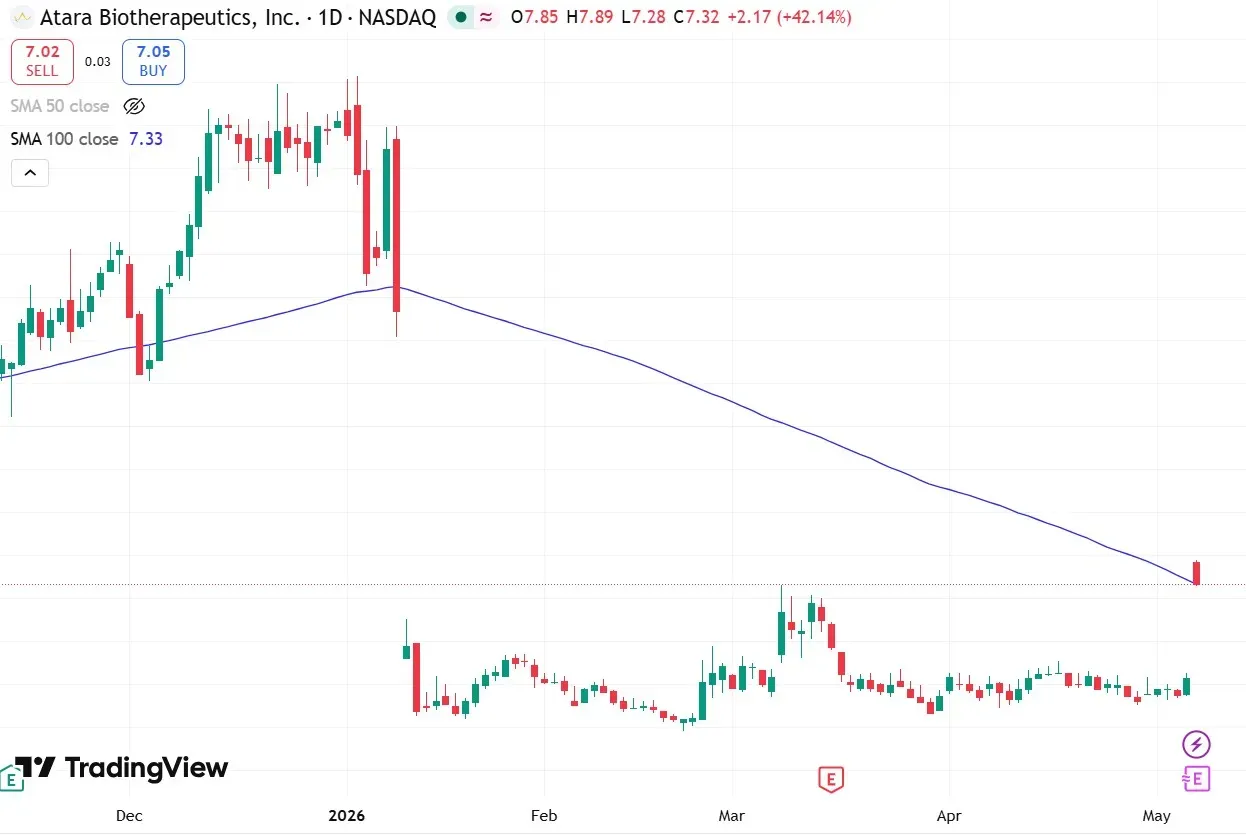
Shares of Atara Biotherapeutics (ATRA) surged more than 43% in morning trade on Thursday, recording their biggest single-day gains in more than two years, after the biotech firm announced a promising update from the U.S. Food and Drug Administration (FDA) tied to its experimental blood cancer therapy.
ATRA shares also breached the 100-day moving average (100-DMA) for the first time in nearly four months.
FDA Talks Signal Resubmission Hopes For ATRA’s Immunotherapy
Atara said it made progress with the FDA on a potential path to resubmitting the biologics license application (BLA) for Tabelecleucel. The drug is used for the treatment of Epstein-Barr virus-associated post-transplant lymphoproliferative disease (EBV+ PTLD).
The update followed a recent Type A meeting between the FDA, Atara, and its partner, Pierre Fabre Pharmaceuticals, after the agency issued a Complete Response Letter (CRL) earlier this year, stating that it could not approve the application in its current form.
However, the FDA has now stated that a single-arm study using a historical control group could support a future marketing application. Pierre Fabre plans to include updated results from its Phase 3 study, including additional patient data and longer follow-up, as part of the resubmission package.
Atara and Pierre Fabre are jointly developing the drug.
Why Did FDA Reject ATRA’s Initial Application?
The FDA previously declined to approve Tabelecleucel, stating the company’s study did not provide enough evidence for accelerated approval and raised concerns about the trial design and analysis.
Pierre Fabre responded to the agency, providing additional explanations and arguing that the study was properly controlled and suitable to support the application. The company also submitted updated long-term efficacy data, along with supportive development and European post-marketing data, for a potential resubmission.
Retail Skeptical About ATRA’s Rally
Retail sentiment on Stocktwits turned ‘extremely bullish’ from ‘neutral’ a day earlier, amid ‘extremely high’ message volumes.
However, one user stated the news did not justify the rally.
Another user called ATRA’s chart setup “neutral” and saw support between $6.50 and $7.
ATRA shares have been under heavy selling pressure this year, slumping nearly 60%.
Read also: IBRX Bulls Shrug Off Earnings Loss -- Retail Traders Eye A Crucial FDA Deadline
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_battery_storage_manufacturing_representative_image_resized_jpg_89a8761f3e.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_stock_chart_rising_resized_6ebc3dd7e4.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_microstrategy_michael_saylor_resized_9fd19e69ec.jpg)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2231786706_jpg_5f9940e890.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_robinhood_bitcoin_OG_jpg_fbd13fff2e.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_COST_jpg_a30a9f74a9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Chinmay_Rautmare_Photo_jpeg_96dd37efd0.webp)