Advertisement|Remove ads.
HIMS Stock's Rally May Have Plenty Of Room Left: FDA Opens Door To Testosterone Therapy

- The FDA signaled openness to expanding testosterone therapy use for low libido linked to idiopathic hypogonadism, a condition without a known cause.
- Hims began building its testosterone-care platform in September 2025, allowing providers to prescribe enclomiphene-based treatment and combination therapy.
- BofA turned bullish on Hims, citing potential upside from upcoming July FDA advisory committee meetings reviewing peptide substances for inclusion on the 503A Bulks List.
Shares of Hims & Hers Health, Inc. (HIMS) have been riding a strong rally on peptide and weight-loss treatment momentum, but a fresh signal from the U.S. Food and Drug Administration (FDA) suggests the telehealth firm’s testosterone therapy could be emerging as the company’s next major growth pillar.
HIMS stock jumped over 11% on Thursday, logging its fourth straight session of gains. Shares are headed for their best week in over a month.
FDA Opens Door To Broader Testosterone Therapy Use
The FDA said on Thursday that it is encouraging sponsors of approved testosterone replacement therapy (TRT) products to contact the agency if they are interested in pursuing a potential new indication for treating low libido in men with idiopathic hypogonadism, a form of low testosterone without a known cause.
The agency said the development follows a preliminary review of published clinical literature and a discussion at a December 2025 expert panel meeting, which identified evidence suggesting TRT may be safe and effective for certain men in this population.
However, sponsors would still need to submit “substantial evidence” of clinical data showing that the benefits outweigh risks, and the FDA has asked interested companies to initiate discussions by April 30.
Currently, FDA-approved TRT therapies are indicated only for men with hypogonadism linked to identifiable medical causes. TRT is used to restore hormone levels in men with low testosterone, a condition that can affect energy, mood, libido, bone density, and muscle strength, and is typically delivered through injections, gels, patches, or oral formulations.
Hims’ Testosterone Care Push
Hims said in September 2025 that providers on its platform can prescribe personalized doses of compounded enclomiphene, a treatment that helps the body restore natural testosterone levels without affecting fertility. For men experiencing sexual-health symptoms linked to low testosterone, the company also introduced combination treatment options that pair enclomiphene with tadalafil, a commonly used erectile dysfunction medication.
The company had also announced plans to expand further into testosterone care this year through exclusive access to the approved oral testosterone therapy Kyzatrex under a partnership with Marius Pharmaceuticals, with injectable testosterone options expected to follow.
Wall Street Flags Peptide Policy Tailwind
The testosterone catalyst is emerging alongside regulatory developments in peptide access, which analysts say could drive additional growth opportunities for the company’s platform.
Bank of America Securities (BofA) recently raised its price target on Hims & Hers, citing potential upside from upcoming FDA advisory committee meetings in July that will review peptide substances for possible inclusion on the federal 503A Bulks List. A favorable outcome could allow the company to repurpose its existing GLP-1 infrastructure for additional peptide therapies as it transitions to branded weight-loss medicines, BofA said.
Separately, Leerink called the recent regulatory progress around peptide access a “clear positive” for Hims & Hers, saying any easing of restrictions could help “fill the growth hole” expected as the company shifts away from compounded obesity treatments. The brokerage added that a favorable advisory committee recommendation could allow Hims & Hers to leverage its existing peptide facility.
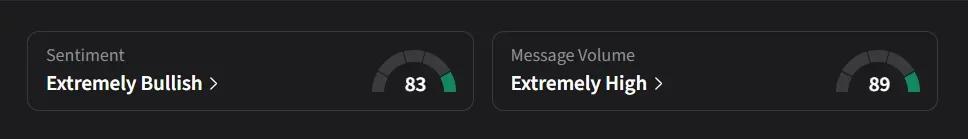
How Do Retail Traders Feel About HIMS?
On Stocktwits, retail sentiment was ‘extremely bullish’ amid nearly a 700% surge in message volumes over the past week.
One user said, “Everyone is excited for a long run like previous ones to $70. Given the momentum, FDA peptide reschedule, Novo partnership, international market broadening, potential short squeeze and earnings in a month, it is going to be a hell of a rally. Huge upside potential !!”
Another user said, “Tons of upside left here…too much good news and more possible revenue for HIMS.”
HIMS stock has just turned positive for the year and is now up about 0.2%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: LCID, ABT, ASBP Stocks Hit 52-Week Lows Today: What’s Driving The Selloff?

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201127733_jpg_293396ee32.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269243885_jpg_4cc426aadd.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/stellarxlm_resized_png_d53f400012.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2158855100_jpg_387300b523.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1243325483_jpg_893f1e0b6b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Boeing_jpg_48e7f3f6e3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)