Advertisement|Remove ads.
IBRX Stock Climbs Overnight: ImmunityBio Declares ‘All Systems Go’ For Dunkirk Anktiva Manufacturing Push

- CEO Richard Adcock said the long-delayed Dunkirk manufacturing site is now “all systems go” after a unanimous resolution at a CCIDA board meeting.
- The Dunkirk facility is expected to serve as a key U.S. production hub for Anktiva.
- ImmunityBio has already invested over $100 million into the project.
Advertisement|Remove ads.
Shares of ImmunityBio, Inc. (IBRX) rose 1% in overnight trading on Wednesday after CEO Richard Adcock said that the company’s long-delayed Dunkirk manufacturing project is now “all systems go,” after local officials unanimously backed progress for the site supporting production of its cancer immunotherapy, Anktiva.
IBRX stock slid nearly 5% to end at $6.96 on Tuesday. However, shares rose 1% in extended trading.
Anktiva US Production Site Back On Track
Following a unanimous consent resolution at a meeting of the Chautauqua County Industrial Development Agency (CCIDA) on Tuesday, ImmunityBio said its long-delayed Dunkirk facility is now moving forward as a key U.S. manufacturing hub for Anktiva production.
Advertisement|Remove ads.
CEO Richard Adcock said that progress had previously stalled after parts of the buildout were left incomplete when a contractor entered bankruptcy, delaying installation and certification required to activate the site.
“Unfortunately, the original contractor didn’t finish parts and other parts didn’t get done. They got trapped in that bankruptcy, and so we all got kind of stuck in limbo land,” Adcock said. “Now that all that’s in our rear view mirror, we can just focus going forward… it’s really now all systems go.”
Adcock said that ImmunityBio has already invested over $100 million into the project and expects to deploy “tens to hundreds of millions” more as the facility moves towards operational readiness. The company has already begun hiring for the restart, identifying at least 19 roles spanning engineering, scientific, and quality-control functions. “These are going to be people with master’s and PhD-level positions… everything is being built out now that we’ve effectively been able to get ourselves untrapped in this situation,” he said.
Advertisement|Remove ads.
A key milestone now in focus is the installation of $50 million in high-speed fill-finish equipment, followed by regulatory certification before engineering runs and initial manufacturing operations can begin.
“It’s not just us getting it ready and running it. It’s then also bringing in the FDA, bringing in the state, bringing in everybody else that regulates it to make sure they’ll certify it,” Adcock said.
Dunkirk Lease Deal Clears Path For Anktiva
Momentum at the Dunkirk site also follows a revised lease agreement with New York State that replaces the earlier $1 lease arrangement under the former Athenex project, which ended after the company went bankrupt in 2023.
Advertisement|Remove ads.
In February, ImmunityBio completed its acquisition of the leasehold interest in the ISO Class 5 pharma manufacturing facility in Dunkirk from Athenex, which includes upstream and downstream processing, fill-finish capability and large-scale lyophilization capacity to produce Anktiva in the U.S.
Under the revised lease terms, the company will pay $525,000 in annual rent, commit at least $40 million toward facility improvements and hire at least 100 employees by the end of 2028, with the workforce expected to expand to 450 by 2032.
The Anktiva Bull Case
The Dunkirk manufacturing push comes as Anktiva continues to drive ImmunityBio’s revenue growth and international expansion strategy. “Since we started this project, we were approved with Anktiva in zero countries. As we sit here today, it’s approved in 34 countries and territories,” Adcock said.
Advertisement|Remove ads.
Preliminary net product revenue for Anktiva hit $44.2 million in the first quarter, up 168% from a year earlier, while full-year 2025 net product revenue rose to $113 million, up 700% from 2024.
Anktiva recently secured conditional authorization across 27 European Union member states, plus Iceland, Liechtenstein and Norway, and also recently became commercially available in Saudi Arabia. The company is also pushing additional programs evaluating Anktiva across lung, pancreatic, colorectal and brain cancers, alongside studies in severe pneumonia, sepsis, long COVID and acute respiratory distress syndrome.
How Do Retail Traders Feel About IBRX?
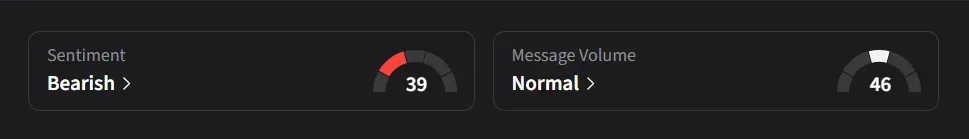
On Stocktwits, retail sentiment for IBRX has slipped to ‘bearish’ from ‘bullish’ levels a week ago amid a 242% surge in message volumes over the past month.
Advertisement|Remove ads.
One user said, “Major! Massive NEWS coming! Slingshot to new highs! Dunkirk and beyond!”
Another user said, “Ahead of plan on Dunkirk hiring! Unanimous approval to company plan and subsidies. BIg checks flowing out- Bigger money coming in soon.....”
Advertisement|Remove ads.
IBRX stock has jumped 165% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Advertisement|Remove ads.
Read Next: LCID, SNGX, SRAD Stock Hit 52-Week Lows Today: What's Driving The Selloff?
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2217856934_jpg_29efdc61ed.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263602546_jpg_a0f4e7e3ca.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2071907975_jpg_85e059f13e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2200882557_jpg_53f3e467bc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2238077141_jpg_984e46421c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_nyse_OG_2_jpg_0e4e1c68f2.webp)