Advertisement|Remove ads.
Can IBRX Stock Snap Its 4-Day Slide? Founder Promises ‘Game-Changer’ Anktiva Data As FDA Speeds Drug Reviews

- Founder Patrick Soon-Shiong said new data tied to the company’s IL-15 cancer immunotherapy platform will soon be submitted to regulators.
- Soon-Shiong said the data relates to Anktiva, describing the IL-15 immune mechanism as a long-recognized “game changer” in cancer treatment.
- He also said he hopes FDA Commissioner Marty Makary reviews the upcoming data closely.
Shares of ImmunityBio Inc. (IBRX) fell over 3% in premarket trading on Monday, despite founder Patrick Soon-Shiong saying that new data tied to the company’s IL-15 cancer immunotherapy platform will soon be submitted to regulators.
IBRX stock extended its losing streak to a fourth consecutive session on Friday, when shares declined more than 3% to close at $8.67.
Founder Signals Upcoming Data Submission
Soon-Shiong said on X that additional data related to Anktiva, the company’s IL-15–based immune-activating therapy, will soon be submitted, describing the IL-15 mechanism as a long-recognized “game changer” in cancer research.
He said institutions including the National Cancer Institute, National Institutes of Health (NIH) and the U.S. Food and Drug Administration (FDA) had recognized IL-15’s potential to stimulate immune responses against tumors since 2007.
Soon-Shiong also said he hopes FDA Commissioner Marty Makary reviews the data closely once it is submitted. “Saving patients' bladders matters. Prolonging life matters. Quality of life matters. T cell and NK cell expansion matters. Duration matters,” he said.
Founder Cites Support For Anktiva’s Mechanism
In a separate post, Soon-Shiong cited comments from former FDA associate commissioner Peter Pitts and NIH Director Jay Bhattacharya, saying they acknowledged that the mechanism of action described in Anktiva’s approved labeling supports the therapy’s scientific rationale.
Soon-Shiong argued that the FDA’s growing emphasis on mechanism-based review could help accelerate patient access to therapies such as Anktiva.
“This is what we have been presenting to the FDA for the past 6 months to desperately allow the 13,000+ requests from Americans to access Anktiva via this pathway,” Soon-Shiong said.
The comments followed Soon-Shiong’s recent appearance on a television interview with Chris Cuomo on NewsNation, where he discussed policy developments and patient access to new cancer therapies.
FDA Pushes Faster Drug Reviews
The remarks also come as the FDA has been signaling a push to accelerate review timelines for certain therapies. Makary recently highlighted on X the agency’s approval of a lung cancer treatment 44 days after filing, describing the process as moving at an “Operation Warp Speed” pace to bring promising therapies to patients more quickly.
The agency has also reportedly launched initiatives to speed up drug reviews and recruit additional scientific staff to address regulatory backlogs and meet review deadlines.
Anktiva’s Regulatory Path Forward
ImmunityBio’s lead therapy, Anktiva, is designed to stimulate immune responses by activating natural killer cells and T cells, enabling the body to mount a stronger response against tumors.
The therapy is currently approved in the U.S. for patients with BCG-unresponsive carcinoma in situ, an aggressive form of bladder cancer that no longer responds to standard BCG treatment. ImmunityBio is now seeking to expand the drug’s approval to include BCG-unresponsive papillary bladder cancer, which represents a significantly larger patient population.
The company previously received a refusal-to-file letter from the FDA for its application seeking that expanded indication and has said it plans to resubmit a biologics license application by the fourth quarter.
How Did Stocktwits Users React?
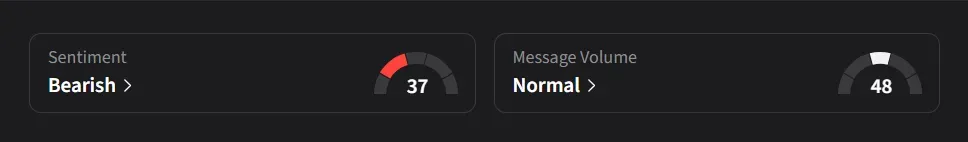
On Stocktwits, retail sentiment for IBRX was ‘bearish’ amid a 67% decrease in message volumes over the past week.
One user said, “Feeling very bullish. I’m here for the long game. Waiting for some more catalysts and will be buying dips as always! Just remember all the institutions that are invested here!!”
Another user urged traders to “lock and load for this week.”
IBRX stock has surged over 330% year-to-date.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2158855100_jpg_387300b523.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1243325483_jpg_893f1e0b6b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Boeing_jpg_48e7f3f6e3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spacex_og_jpg_690b281532.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201736876_jpg_77932bc771.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2271060260_jpg_b0369496dd.webp)