Advertisement|Remove ads.
NTLA Stock Surges 35% Overnight: Retail Mood Peaks Ahead Of First-Ever Phase 3 Readout For Gene-Editing Therapy

- The Haelo Phase 3 trial marks the first late-stage test of an in-vivo CRISPR gene-editing treatment.
- Earlier mid-stage studies showed a 96% reduction in monthly attack rates, with 97% of patients attack-free for up to three years.
- The FDA recently lifted clinical holds on two Phase 3 trials of Nex-z, its Transthyretin Amyloidosis program being developed with Regeneron.
Advertisement|Remove ads.
Shares of Intellia Therapeutics, Inc. (NTLA) surged 35% overnight heading into Monday as retail excitement peaked ahead of a Phase 3 readout that could mark the first late-stage validation of an in-vivo CRISPR gene-editing platform.
NTLA stock is coming off from its worst week in nearly three months after sliding 9% last week.
NTLA Nears First Phase 3 In-Body CRISPR Readout
The company said it will report topline results from its global Phase 3 Haelo trial evaluating Lonvo-z on Monday. Lonvo-z is a one-time gene-editing treatment to prevent swelling attacks in patients with hereditary angioedema. The therapy works by switching off the KLKB1 gene, which produces a protein that triggers these attacks.
Advertisement|Remove ads.
Hereditary angioedema is an inherited disorder that causes recurring episodes of severe swelling in areas such as the face, abdomen, and limbs, which can sometimes be life-threatening if breathing is affected.
Earlier mid-stage studies showed reductions in attack frequency, with monthly attack rates falling about 96% from baseline and 97% of treated patients remaining attack-free for up to three years.
Investors are watching the readout closely as the Haelo study marks the first Phase 3 test of an in-body CRISPR gene-editing therapy, in which the treatment will permanently modify a disease-causing gene after a single infusion. If confirmed, the results could make Lonvo-z the first one-time treatment capable of replacing chronic preventive therapy for hereditary angioedema.
Advertisement|Remove ads.
NTLA Clears FDA Holds On Two Phase 3 CRISPR Trials
The upcoming readout positive regulatory progress in another late-stage CRISPR program in recent times. Last month, the U.S. Food and Drug Administration (FDA) lifted a clinical hold on the Magnitude Phase 3 trial evaluating Nex-z for a heart condition caused by Transthyretin protein buildup, allowing enrollment to resume after better safety measures were introduced.
The hold had originally been imposed in October 2025 after a treated patient showed signs of liver enzyme elevations and increased bilirubin levels, pointing to potential liver injury.
Separately, In January, the FDA lifted a clinical hold on the related Magnitude-2 Phase 3 trial evaluating Nex-z in the nerve form of the same disease, enabling enrollment to restart. Nex-z is being developed as a one-time gene-editing treatment to switch off the gene that produces Transthyretin protein and is being advanced in collaboration with Regeneron Pharmaceuticals.
Advertisement|Remove ads.
How Do Retail Traders Feel About NTLA?
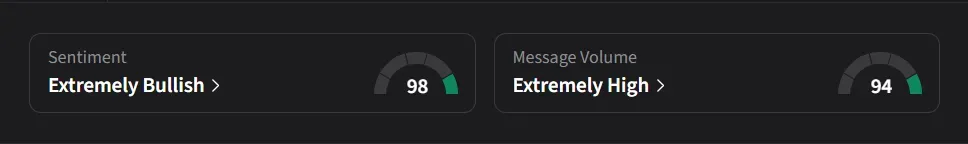
On Stocktwits, retail sentiment for NTLA surged to a record 98/100, shifting to ‘extremely bullish’ from ‘bullish’ levels a week earlier as message volumes jumped nearly 2,000% over the past week.
One user said, “sell the news events are much more likely to occur when the stock has run up over time, in anticipation of the catalyst. That hasn’t happened here”
Advertisement|Remove ads.
Another user said, “Intellia has some big data out Monday! A big result will goose the whole biotech sector in my opinion: ReRating is overdue. CAR-T sector hot…some tailwind is everything”
NTLA stock has jumped 67% over the past year.
Advertisement|Remove ads.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_fuelcellenergy_resized_ef1a57cfe6.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_oscar_health_jpg_89aa74c6fe.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2217651425_jpg_0d7dc8a6cd.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spy_jpg_a85fe2a8bc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2217856934_jpg_29efdc61ed.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263602546_jpg_a0f4e7e3ca.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)