Advertisement|Remove ads.
RCKT Stock Jumps Over 10% Premarket: FDA Approves Its First Gene Therapy For Rare Childhood Immune Disease
- The FDA approved gene therapy Kresladi, Rocket's first commercial product.
- The approval targets children with severe LAD-I lacking a matched sibling donor for a stem-cell transplant.
- Rocket received a Rare Pediatric Disease Priority Review Voucher, which can be used or sold to accelerate another drug review.
Shares of Rocket Pharmaceuticals Inc. (RCKT) jumped 11% in premarket trading on Friday after the U.S. Food and Drug Administration (FDA) approved its gene therapy Kresladi, marking the company’s first commercial authorization and earning it a Rare Pediatric Disease Priority Review Voucher.
RCKT stock rose modestly on Thursday, with shares jumping by more than 13% so far this week. Meanwhile, trading picked up amid the approval, with volume hitting about 8.89 million shares, roughly 3.6x the stock’s average daily volume.
Rocket Wins First Gene-Therapy Approval
Kresladi received accelerated approval for children with severe Leukocyte Adhesion Deficiency Type I (LAD-I), a rare genetic immune disorder, who do not have a suitable matched sibling donor for a stem-cell transplant.
The treatment uses the patient’s own blood stem cells, which are genetically adjusted to carry working copies of the ITGB2 gene. These corrected cells are then returned to the body in a single infusion, helping the immune system function more normally by addressing the underlying cause of the disease.
“Today’s accelerated approval provides a breakthrough treatment for pediatric patients with severe Leukocyte Adhesion Deficiency Type I—the first FDA-approved gene therapy to treat this disease,” said Vinay Prasad, director of the FDA’s Center for Biologics Evaluation and Research (CBER).
The approval was based on improvements in key immune-function markers called CD18 and CD11a, which were seen at 12 months and sustained through 24 months after treatment, indicating better white blood cell activity. Since the therapy received accelerated approval, Rocket must complete follow-up studies to confirm long-term clinical benefit.
FDA Grants Priority Review Voucher
Kresladi previously received Orphan Drug, Rare Pediatric Disease, Regenerative Medicine Advanced Therapy, and Fast Track designations from the FDA, signaling both the seriousness of the condition and the limited treatment options available.
Alongside approval, Rocket was awarded a Rare Pediatric Disease Priority Review Voucher. These vouchers can be used to speed up the review of another therapy or transferred to another company.
Rocket Expands Cardiac Pipeline
Beyond LAD-I, Rocket is also advancing its heart-disease gene-therapy pipeline. The company expects to treat more patients in a key mid-stage study of Danon disease in the first half of 2026, begin an early-stage study of dilated cardiomyopathy later this year, and continue discussions with the FDA on plans for a late-stage trial targeting arrhythmogenic cardiomyopathy.
As of Dec. 31, 2025, Rocket reported $188.9 million in cash, cash equivalents and investments. The company expects to use the funds for operations into the second quarter of 2027, excluding any potential proceeds from the newly awarded priority review voucher.
What Is The Retail Mood On Stocktwits?
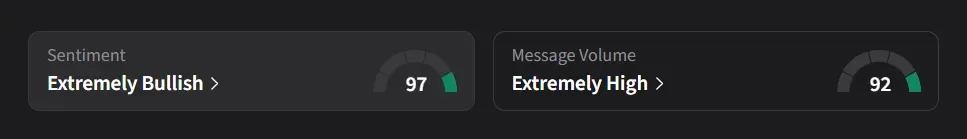
On Stocktwits, retail sentiment for RCKT jumped to ‘extremely bullish’ from ‘neutral’ levels since the FDA approval amid an eye-watering 8,600% surge in message volumes this quarter.
One user expects the RCKT stock to hit “$8-$9 by end of day. $7 minimum.”
Another user noted, “Priority review voucher alone is worth $2 or $200 million. Let alone approval.”
RCKT stock has jumped 34% so far this year, but down nearly 40% over the past 12 months.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_kevin_warsh_OG_3_jpg_bbccdc89d7.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2241231078_jpg_b6bbd51e0c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_6979_jpg_a2a1032fdc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Kevin_Warsh_Donald_Trump_jpg_4567f56ac5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2253111144_jpg_12be2fed14.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Jensen_Huang_jpg_8579a8a271.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/AMD_s_Lisa_Su_1200pi_gettyimages_resized_jpg_2b6947251d.webp)