Advertisement|Remove ads.
IBRX Stock Rises After NCCN Guideline Update Expands ANKTIVA’s Use - Retail Expects Sales To Surge

- The NCCN updated its 2026 bladder cancer guidelines to include ANKTIVA in combination with BCG for patients with BCG-unresponsive non-muscle invasive bladder cancer with papillary-only disease.
- The earlier guideline only included ANKTIVA for patients with BCG-unresponsive NMIBC with CIS with or without papillary disease.
- ANKTIVA’s use for papillary-only disease has not yet been approved by the FDA.
Advertisement|Remove ads.
Shares of ImmunityBio Inc. (IBRX) gave up most of their early gains to trade 1% higher on Tuesday, after a key update to its U.S. cancer treatment guidelines expanded the recommended use of its lead therapy ANKTIVA in bladder cancer.
The National Comprehensive Cancer Network (NCCN) has updated its 2026 bladder cancer guidelines to include ANKTIVA in combination with Bacillus Calmette-Guérin (BCG) for patients with BCG-unresponsive non-muscle invasive bladder cancer with papillary-only disease.
In the NCCN guidelines, ANKTIVA is listed alongside other bladder-sparing options like chemotherapy and gene therapy, while surgery and clinical trials remain the preferred treatments.
Advertisement|Remove ads.
What Changed In NCCN Guidelines?
The earlier NCCN guidelines only included ANKTIVA for patients with BCG-unresponsive NMIBC with carcinoma in situ (CIS) with or without papillary disease. The treatment boosts natural killer and T cells to fight cancer and has shown lasting benefits in studies. However, its use for papillary-only disease has not yet been approved by the U.S. Food and Drug Administration (FDA).
ANKTIVA works by activating natural killer (NK) and T cells, helping the immune system target cancer more effectively.
“The addition of ANKTIVA plus BCG for papillary-only disease in the NCCN guidelines reflects the growing body of clinical data evaluating ANKTIVA in this patient population and reinforces our commitment to developing comprehensive treatment solutions that address the full spectrum of patients living with BCG-unresponsive NMIBC,” said Patrick Soon-Shiong, M.D., Founder, Executive Chairman and Global Chief Scientific and Medical Officer of ImmunityBio.
Advertisement|Remove ads.
How Did Stocktwits Users React?
Retail sentiment for IBRX on Stocktwits remained in the ‘bearish’ territory over the past 24 hours.
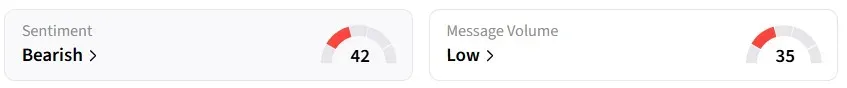
One user called the updated NCCN guidelines a “game changer” for the company and expects sales to surge.
Advertisement|Remove ads.
Another user said NCCN recommendations carry “significant weight” in oncology practice and are often used by payers to support coverage even without FDA approval.
Year-to-date, IBRX shares have gained more than 310%.
Advertisement|Remove ads.
Read also: OKLO Stock Surges Into Retail Spotlight After Key Nuclear License Win
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_strategy_logo_OG_jpg_fa4e1a7d04.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1376765653_jpg_d56a66290e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275170769_jpg_e811c3ae51.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spacex_jpg_6eb3abcb72.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2271060260_jpg_b0369496dd.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Marvell_jpg_2d5ebc76bc.webp)