Advertisement|Remove ads.
ARDX Stock Jumps Over 5% Overnight As Director Loads Up In Vote Of Confidence For Flagship Constipation Drug

- Director David Mott bought 333,333 shares at a weighted average price of $5.84, lifting his stake to more than 3.3 million shares.
- The insider buy followed a mixed Q4 earnings report in which revenue rose to $125.2 million, beating expectations, while earnings remained flat.
- Ibsrela drove the revenue beat, offsetting softer Xphozah sales, and the company guided for Ibsrela revenue of $410 million to $430 million in 2026.
Advertisement|Remove ads.
Ardelyx shares jumped more than 5% overnight after a board director made a sizable open-market purchase, adding fuel to growing investor confidence in Ibsrela, the company’s flagship constipation drug.
ARDX stock rose 0.4% on Monday after tumbling 15% on Friday, when the company reported no earnings for the fourth quarter (Q4).
Ardelyx Director Buys 333K Shares In Open Market
According to a new regulatory filing, David Mott, a director at Ardelyx, purchased 333,333 shares of the company’s stock on Friday at a weighted average price of $5.84 per share.
Advertisement|Remove ads.
The filing shows the purchase was made in multiple open-market transactions, with prices ranging from $5.67 to $6.00. Following the buy, Mott holds over 3.3 million Ardelyx shares.
Ibsrela Drives Q4 Revenue Beat
The insider buy followed Ardelyx’s Q4 earnings report, where revenue rose to $125.2 million, up from $116.1 million a year earlier. The result topped Wall Street expectations of about $119.4 million.
The company said the increase was driven largely by higher adoption of Ibsrela, which is approved for the treatment of irritable bowel syndrome with constipation (IBS-C) in adults. Growth in Ibsrela helped offset softer sales from Xphozah, Ardelyx’s drug for lowering phosphate levels in patients with chronic kidney disease on dialysis.
Advertisement|Remove ads.
Earnings per share came in at $0.0, compared with $0.02 in the year-ago quarter and slightly below expectations of $0.01.
Strong 2026 Outlook Anchored By Ibsrela
Alongside the results, Ardelyx expects Ibsrela revenue of $410 million to $430 million in 2026, representing growth of at least 50% from 2025 levels. The company cited improvements in prescribing across target healthcare providers, higher patient engagement, and stronger prescription pull-through. For Xphozah, the company forecast 2026 revenue of $110 million to $120 million, up from $103.6 million in 2025.
Ardelyx also said it is running additional programs to evaluate Ibsrela in chronic idiopathic constipation and continues to advance its experimental asset RDX10531 across multiple potential indications.
Advertisement|Remove ads.
Analysts Reaffirm Bullish Views
On Monday, Wedbush maintained its $19 price target on Ardelyx shares, implying a 230% upside from current levels. Meanwhile, H.C. Wainwright raised its target by 80% to $18 from $10 on Friday, while reiterating a ‘Buy’ rating.
H.C. Wainwright said it was encouraged by Ardelyx securing a new U.S. patent extending to November 2042, covering oral formulations of Tenapor, including Ibsrela and Xphozah. Ardelyx has since confirmed that both drugs are now listed in the FDA’s Orange Book, marking a step that strengthens intellectual property protection.
CEO Mike Raab previously said the listing was “an important step in extending protections for our portfolio of first-in-class medicines.”
Advertisement|Remove ads.
How Did Stocktwits Users React?
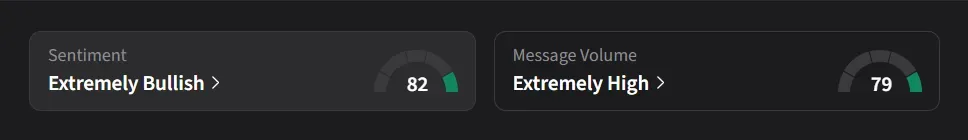
On Stocktwits, retail sentiment for ARDX was ‘extremely bullish’ amid ‘extremely high’ message volume.
One user said, “Maybe retailers will realize now’s the time to buy. Nice buy by mott ”
Advertisement|Remove ads.
Another user said, “Always nice seeing Mr.Mott’s investing in the company. Clearly, he’s in it to win it. He knows what he owns and how the current strategy will be a success. So swell! “
ARDX stock has declined 1.4% year to date.
Advertisement|Remove ads.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1398540998_jpg_aa1a77f11b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2247874160_jpg_4fb51355e6.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Elon_Musk_Space_X_jpg_4284182546.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2240006388_jpg_320990af67.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2208941052_jpg_64e32eb34c.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/gettyimages_2274057958_594x594_jpg_66274b07c8.webp)