Advertisement|Remove ads.
Capricor Or Sarepta: Two DMD Stocks Enter A High-Stakes Week – Which One Has More Upside?

- Capricor is preparing a February submission to the FDA for its DMD cell therapy Deramiocel, following a formal request for the full Phase 3 Hope-3 clinical study report.
- Koyfin expects Sarepta to post Q4 revenue of $390.95 million, and an EBITDA loss of $144.34 million.
- Koyfin estimates call for no revenue, and an EBITDA loss of $24.60 million for Capricor.
Shares of Capricor Therapeutics, Inc. (CAPR) and Sarepta Therapeutics, Inc. (SRPT) are in focus this week as investors weigh different near-term catalysts tied to Duchenne muscular dystrophy (DMD), a severe genetic disorder that causes progressive muscle degeneration and heart failure, primarily in young boys.
Capricor is preparing a regulatory filing with the U.S. Food and Drug Administration (FDA) and is also set to report fourth-quarter (Q4) earnings this week, while Sarepta reports Q4 results amid safety and legal overhangs.
Capricor Prepares February FDA Submission For Deramiocel
Capricor is positioning for a February submission to the FDA related to its cell therapy Deramiocel, following a formal request from the agency for the full clinical study report from the Phase 3 Hope-3 trial. The company said preparation of the report is “well underway” and that the submission is intended to address the issues raised in a Complete Response Letter issued in July 2025.
Importantly, the FDA did not request any new clinical trials or additional patient data as part of the request. The Hope-3 trial enrolled 106 boys and young men with Duchenne muscular dystrophy, including patients who can no longer walk, and met its primary endpoint by showing a slowing in the decline of upper limb function.
The study also demonstrated improvement in a key cardiac endpoint measuring left ventricular ejection fraction. Capricor said the safety profile was consistent with earlier studies.
Wall Street View On CAPR
Capricor is scheduled to report quarterly results on Friday. According to Koyfin estimates, the company is expected to report no revenue for the quarter, with earnings before interest, taxes, depreciation, and amortization (EBITDA) projected at a loss of $24.60 million, compared with a loss of $25.79 million last quarter.
Earnings before interest and taxes (EBIT) are expected at a loss of $25.87 million versus $26.28 million last quarter. Adjusted EPS is projected at a loss of $0.55, widening slightly from $0.54 in the previous quarter.
Analyst sentiment around Capricor remains strongly following the Phase 3 data. Koyfin data show a 12-month average price target of $50.8, implying a 78% upside from the stock’s last close. Of the 10 analysts covering the stock, two rate it ‘Strong Buy’ and eight rate it ‘Buy.’
Sarepta Faces Earnings Amid Legal and Safety Overhangs
Sarepta Therapeutics is due to report fourth-quarter (Q4) results after market close on Wednesday. The earnings release comes as the company continues to navigate regulatory scrutiny of its Duchenne gene therapy, Elevidys, and renewed legal uncertainty after the U.S. Court of Appeals for the Federal Circuit reinstated a patent lawsuit brought by Regenxbio.
The lawsuit centers on claims that Elevidys infringes gene-therapy patents licensed from the University of Pennsylvania, with Regenxbio and Penn seeking more than $900 million in damages. Sarepta has also faced safety concerns after two patients who could not walk died from acute liver failure following Elevidys treatment in 2025. While shipments have resumed for ambulatory patients, the company is still developing a risk-mitigation strategy to resume use in non-ambulatory patients.
Wall Street View On SRPT
Koyfin estimates show Sarepta is expected to report quarterly revenue of $390.95 million, down from $399.36 million in the prior quarter. EBITDA is projected to widen to a loss of $144.34 million from $91.17 million previously, while EBIT is expected at a loss of $136.41 million versus $103.39 million. Adjusted EPS is expected at a loss of $0.77 versus an earlier $0.13.
Analyst sentiment on Sarepta is notably more mixed than on Capricor. Koyfin data shows a 12-month average price target of $21.43, implying a 14% upside from the stock’s current levels. Among 26 analysts covering the stock, seven rate it ‘Buy’, fourteen rate it ‘Hold’, three rate it ‘Sell’, and two rate it ‘Strong Sell’.
How Did Stocktwits Users React?
On Stocktwits, retail sentiment toward Capricor was ‘extremely bullish’ amid ‘extremely high’ message volume, while sentiment toward Sarepta was ‘bearish’ amid ‘low’ message volume.
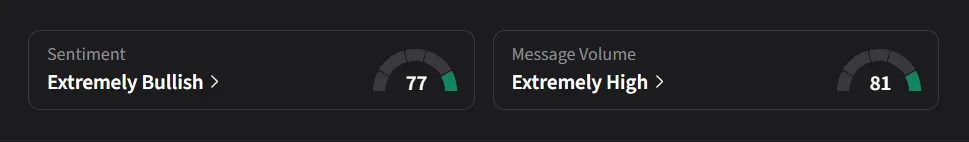
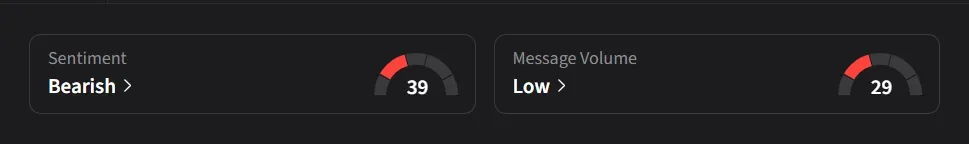
One user said they were “patiently waiting for the Roche buyout” of Capricor.
Another user said, “Serapta can make much safer one for DMD. We will see the development very soon!”
While CAPR stock has declined 1.2% year-to-date, SRPT stock slid 12% over the same period.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_PVH_c4c68472c9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_6979_jpg_a2a1032fdc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_crowdstrike_resized_jpg_ae45d5de0e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2239579889_jpg_526b21945a.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2268516558_jpg_8245ea1920.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Broadcom_jpg_f302b01f15.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)