Advertisement|Remove ads.
CMPS Stock Surges As White House Psychedelic Push Prompts FDA Review, Boosts COMP360 Prospects

- The order calls on the FDA to give priority to select psychedelic treatments that have demonstrated positive results in preliminary research.
- The treatments could qualify for priority review, enabling regulators to assess them faster while upholding safety requirements.
- COMP360, Compass’s psilocybin therapy in late-stage trials, has shown relief within a day, with effects lasting up to six months after one or two doses.
Compass Pathways (CMPS) stock gained overnight on Sunday as the company voiced support for the White House’s executive action to speed up research, approval, and availability of psychedelic-based treatments.
The company specializes in psilocybin therapies, which stand to benefit from faster regulatory timelines tied to its lead candidate.
Regulatory Changes To Speed Access
President Donald Trump on Saturday signed the order to speed up the development, review, and accessibility of treatments targeting complex psychiatric conditions. The policy outlines a coordinated effort across federal agencies to reduce regulatory delays and expand research opportunities. The order instructs the Food and Drug Administration (FDA) to prioritize certain psychedelic drugs that have already shown promise in early studies.
These therapies may receive special review vouchers, allowing regulators to evaluate them more quickly without compromising safety standards. In parallel, the FDA and the Drug Enforcement Administration are tasked with designing pathways that permit eligible patients to access investigational treatments still under review, provided they meet baseline safety criteria.
Compass Lead Candidate Gains Momentum
Compass said it is already engaged in ongoing review discussions with the FDA, positioning it to capitalize on the policy shift as it advances treatment for depression.
“Compass is conducting the largest, most robust classic psychedelic studies to date with COMP360 synthetic psilocybin,” said CEO Kabir Nath.
At the center of this development is COMP360, the company’s synthetic psilocybin therapy currently in late-stage trials. Phase 3 trial findings suggest the treatment may produce rapid improvements, with patients experiencing relief within a day and sustained benefits lasting about six months after just 1 or 2 doses.
Compass Pathways’ stock was up over 2% as of its last after-hours close. Peers Enveric Biosciences and ATAI Life Sciences were up over 3.5% and 1.1%, respectively.
The company’s study includes data from more than 1,000 participants, indicating a generally manageable safety profile with short-lived side effects. Beyond depression, Compass is expanding its focus to PTSD, including collaboration with Veterans Affairs programs to explore treatment options.
What Are Retail Traders Saying?
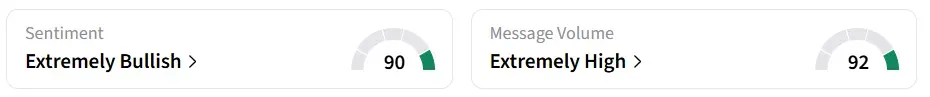
On Stocktwits, retail sentiment for Compass Pathways remained in ‘extremely bullish’ territory amid ‘extremely high’ message volume levels, which have surged by 2,000% over the past seven days.
A Stocktwits user expressed optimism about COMP360 receiving approval and availability. “EO [executive order] was the last hurdle in my mind for this stock. Federal govt has cleared the run way for psychedelic medicine and the FDA is on board. I truly believe this will be approved and is going to market. This is the future of mental health.”
Another user said: “This is not a sell the news event. This news was not expected. This will skyrocket upon FDA approval anticipation. Once FDA approved, I could see a sell the news event but for now enjoy the gains this week.”
CMPS stock has declined over 3% year-to-date.
Also See: Apple’s China Surge Shows Pricing Power Matters In A Supply Crunch – Retail Bulls Take Notice
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1233265488_jpg_60af97169a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275851163_jpg_b115c861be.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2237640344_jpg_bc97a7240c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2227347116_jpg_6a738ad597.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201127733_jpg_293396ee32.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269243885_jpg_4cc426aadd.webp)