Advertisement|Remove ads.
Gossamer Bio Stock Plummeted 82% Today — What’s Behind The Crash?
- Participants treated with seralutinib recorded a median improvement of 28.2 meters in walking distance, compared with 13.5 meters with placebo.
- The estimated treatment benefit was 13.3 meters, with a p-value of 0.032, falling short of the predefined significance cutoff of 0.025.
- In a predefined subgroup of intermediate- and high-risk individuals, seralutinib delivered a more pronounced benefit.
Gossamer Bio Inc. (GOSS) on Monday released topline findings from its late-stage PROSERA trial evaluating seralutinib in patients with pulmonary arterial hypertension (PAH), offering a mixed picture as the company weighs its regulatory strategy.
The Phase 3 study assessed the experimental therapy over 24 weeks, measuring improvement in six-minute walk distance compared with placebo.
Primary Endpoint Falls Short
The drug did not achieve the predefined statistical bar for its main goal. Participants treated with seralutinib recorded a median improvement of 28.2 meters in walking distance, compared with 13.5 meters among placebo recipients.
The estimated treatment benefit was 13.3 meters, with a p-value of 0.032, falling short of the predefined significance cutoff of 0.025. As a result, the secondary endpoints cannot be formally considered statistically significant, though all four key secondary measures favored seralutinib.
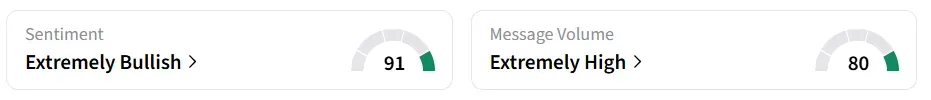
Following the announcement, Gossamer Bio's stock traded over 82% lower on Monday mid-morning. On Stocktwits, retail sentiment around the stock remained in ‘extremely bullish’ territory while message volume shifted to ‘extremely high’ from high’ levels in 24 hours.
Stronger Results In Higher-Risk Patients
In a predefined subgroup of intermediate- and high-risk individuals, seralutinib delivered a more pronounced benefit. That cohort achieved a 20-meter placebo-adjusted increase in walking distance, along with favorable results across most secondary markers.
Patients with connective tissue disease–associated PAH also demonstrated a notable response, with a placebo-adjusted increase of 37 meters in walking distance at 24 weeks.
The company plans to meet with the U.S. Food and Drug Administration to determine a regulatory path forward. Gossamer has paused enrollment in SERANATA study to assess the implications of PROSERA’s outcome.
GOSS stock has declined by over 70% in the last 12 months.
Also See: Why Did Allurion Stock Surge Over 50% Today?
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2239579889_jpg_526b21945a.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2268516558_jpg_8245ea1920.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Broadcom_jpg_f302b01f15.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2227669377_jpg_9a115c3623.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2277830109_jpg_8566757c70.webp)