Advertisement|Remove ads.
HIMS Stock Soars 10% Premarket: BofA Flags Fresh Upside As FDA Peptide Pivot Could Turn GLP-1 Capacity Into New Growth Engine

- The FDA scheduled advisory meetings on July 23-24 to review several peptide substances for possible inclusion on the 503A Bulks List.
- BofA raised its price target in HIMS to $25 from $21, citing potential upside from FDA peptide policy developments.
- The FDA review follows 2023 restrictions on nearly 20 peptides over safety concerns.
Shares of Hims & Hers Health, Inc. (HIMS) jumped over 10% in premarket trading on Thursday after Bank of America Securities (BofA) raised its price target and flagged potential new revenue upside from upcoming FDA talks that could expand access to peptide therapies beyond GLP-1 treatments.
BofA Sees GLP-1 Capacity Repurposed For Peptides
BofA lifted its price target on Hims to $25 from $21, while maintaining a ‘Neutral’ rating. The firm raised its outlook following a recent notice from the U.S. Food and Drug Administration (FDA) that it is scheduling July 23-24 meetings to review several peptide substances for possible inclusion on the 503A Bulks List, which determines which ingredients pharmacies can legally use to prepare customized medicines.
The brokerage said the move could open the door to new potential revenue opportunities for the company. BofA added that the development could allow the company to repurpose existing GLP-1 capacity for other peptide therapies as it transitions its weight-loss strategy toward branded treatments, according to an Investing.com report.
To reflect the additional revenue stream and higher peer multiples, BofA raised its valuation multiple assumption to 25.5x from 21.5x. However, BofA noted that the FDA’s action represents only an initial step, not confirmation that any peptide substances will ultimately be added to the compounding list.
FDA Revisits Peptides Restricted In 2023
The FDA’s upcoming review follows setbacks in 2023, under the Joe Biden administration, to place nearly 20 peptide substances on a federal restriction list preventing their use by compounding pharmacies after advisers concluded the drugs lacked sufficient clinical safety evidence.
Regulators at the time cited risks including potential cancer and liver, kidney, and heart complications, noting that many peptides promoted across wellness markets had not been extensively studied in humans.
U.S. Health and Human Services Secretary Robert F. Kennedy Jr. said on X that the review process is intended to help shift demand away from “a dangerous black market that puts Americans at risk.”
Hims Shifts Toward Branded GLP-1 Treatments
The peptide-linked upside comes as Hims continues shifting its weight-loss business toward FDA-approved branded GLP-1 therapies following its agreement with Novo Nordisk. As part of the deal, the company plans to discontinue marketing compounded GLP-1 alternatives and instead offer Wegovy alongside Ozempic for eligible patients.
CEO Andrew Dudum recently said that the company has already shipped its first Novo Nordisk medicines and is on track to exceed a run rate of 100,000 Wegovy prescriptions per month as part of the rollout.
How Do Retail Traders Feel About HIMS?
On Stocktwits, retail sentiment for HIMS jumped to ‘extremely bullish’ from ‘bearish’ levels a day ago amid over a 400% surge in 24-hour message volumes.
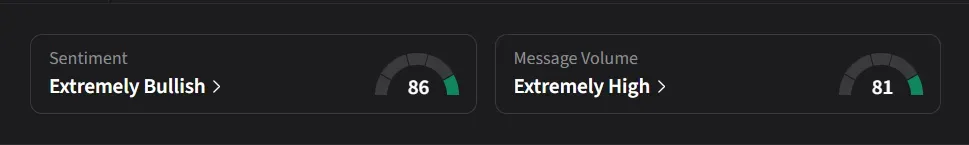
Several users highlighted high short interest in HIMS shares, which currently hovers near a record high of 36.6%, according to Koyfin data.
One user said, “Is $HIMS about to melt faces? Probably. With a 30% short interest and massive momentum, we’re looking at a prime short squeeze setup. These are exactly the high-stakes setups we live for. Let the fireworks begin.”
Another user said, “Interesting day ahead guys. short position must be covered now. too much risk for more positive news to come.”
HIMS stock has declined 18% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2258806047_jpg_085e5f0fe0.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2261740680_jpg_8eee597409.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2204154647_jpg_f2ab9ac6ab.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2191702229_jpg_e9b50f268b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2270340049_jpg_4327dac6f7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/moderna_hq_resized_jpg_97563ed423.webp)