Advertisement|Remove ads.
IBRX Stock Eyes Best Week Since February: Founder Highlights New Anktiva-NK Cell Therapy Study

- IBRX stock extended losses overnight after ImmunityBio reported a Q1 net loss of $632.8 million versus $129.6 million a year ago.
- Founder Patrick Soon-Shiong reignited investor interest after teasing a new Anktiva-based cell therapy trial.
- ImmunityBio said Anktiva demand continued growing across new prescribers and maintenance.
Shares of ImmunityBio, Inc. (IBRX) are heading for their best week in over two months as investors shifted focus from the company’s post-earnings selloff toward a newly teased Anktiva combination cell therapy trial.
IBRX stock has gained 9% so far this week and is on track for its best weekly performance since Feb.27, despite tumbling over 7% on Thursday after the company’s first-quarter (Q1) earnings release.
New Anktiva Combo Trial In Focus
IBRX stock extended its slide in overnight trading on Friday after ImmunityBio reported a Q1 net loss of $632.8 million, compared with $129.6 million a year earlier, while revenue of $44.2 million came in slightly above consensus estimates of $43.9 million.
However, founder Patrick Soon-Shiong attempted to add fresh momentum after posting on X that a Phase 1 trial involving NK-92 cells in refractory lymphoma and multiple myeloma, conducted nearly a decade ago, demonstrated safety and evidence of efficacy, including 2 complete responses. He also said that the company plans to begin a trial combining Anktiva, the company’s flagship immunotherapy, with targeted cell therapy, calling it the “Power of NK and T!”
ImmunityBio Highlights Anktiva Demand
The earnings report still highlighted commercial momentum for Anktiva despite the massive increase in quarterly loss. “We continue to see strong demand for ANKTIVA from both new prescribers and physicians expanding use across multiple eligible patients, including in the maintenance setting,” said Richard Adcock, CEO of ImmunityBio.
He also noted that the company expanded market access internationally, with Anktiva now commercially available in Saudi Arabia and additional launches expected later this year.
ImmunityBio reported Q1 product revenue growth of 168% from the previous year and ended the quarter with $381 million in cash, cash equivalents and marketable securities.
The larger quarterly loss was mainly due to non-cash fair-value changes from warrant liabilities, derivative liabilities and related-party convertible notes, along with higher research, clinical trial and commercialization expenses.
FDA Filing Clock Nears Key Deadline
Another major focus for traders is the FDA’s ongoing review of ImmunityBio’s resubmitted supplemental biologics license application for Anktiva plus standard BCG in patients with a non-muscle invasive bladder cancer with papillary tumors.
The FDA’s standard 60-day filing review window is about to expire, after which the agency determines whether the application is complete enough to formally accept for full review and assign a target action date. The filing includes long-term data from the Quilt 3.032 study, which ImmunityBio said showed that 58.2% of patients remained cancer-free after 12 months and that more than 80% avoided bladder-removal surgery over three years.
Investor attention is also turning toward next week’s American Urological Association Annual Meeting in Washington, where ImmunityBio plans to present additional Anktiva analyses comparing the therapy against rival bladder cancer treatments, including Johnson & Johnson’s TAR-200 and Nadofaragene Firadenovec. Soon-Shiong is also expected to present new data around IL-15 immune activation and broader immunotherapy approaches.
How Do Retail Traders Feel About IBRX?
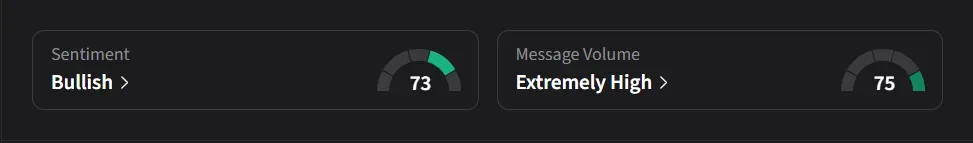
On Stocktwits, retail sentiment for IBRX was ‘bullish’ amid ‘extremely high’ message volume.
One user said, “The IBRX community on Stocktwits is more than just investors — we are believers in the vision behind Anktiva and ImmunityBio. Many of us are committed not only financially, but personally, to helping accelerate the life-saving solutions to the world.”
Another user said, “Combined with the potential for positive FDA news, this could set up a strong move toward new highs, especially since the company is fundamentally very different from where it was 3 or 6 months ago.”
IBRX stock has surged 311% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2249184989_jpg_2f36d82a30.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_950698324_jpg_d6201b2562.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_951165788_jpg_24c6559b2b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1368998806_2_jpg_e532306557.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1163013566_jpg_dc3a81571d.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2229273884_jpg_b009b52b81.webp)