Advertisement|Remove ads.
IBRX Stock Plunges 27% After FDA Flags ‘Misleading’ Promotion Of Bladder Cancer Drug ANKTIVA

- The FDA said the TV ad and podcast wrongly suggest that ANKTIVA can keep all NMIBC patients cancer-free long term.
- The regulator asked ImmunityBio to respond within 15 days and fix the misleading promotions.
- The FDA had warned the company twice before about issues with how ANKTIVA was presented.
Shares of ImmunityBio (IBRX) tanked nearly 27% on Tuesday after the U.S. Food and Drug Administration (FDA) issued a warning letter to the pharmaceutical company, raising concerns about the promotion of its bladder cancer treatment, ANKTIVA.
“These violations are concerning from a public health perspective because the promotional communications create a misleading impression that ANKTIVA, a treatment for a certain type of bladder cancer, can cure and even prevent all cancer,” the letter read.
The FDA had sent warning letters on Sept. 9, 2025, and Jan. 7, 2026, flagging similar issues with how ANKTIVA was presented. Those letters noted the company used data from a single-arm study in a way that could mislead people about the drug’s effectiveness.
FDA Flags ANKTIVA Claim Made In Ad And Podcast
The FDA highlighted a TV advertisement and a podcast featuring CEO Richard Adcock and Executive Chairman Patrick Soon-Shiong, which made strong claims about ANKTIVA’s ability to “treat all cancers.” The regulator said the representations in the TV ad and podcast are “misleading.”
The FDA said that the TV ad and podcast wrongly suggest that ANKTIVA can keep all Non-Muscle-Invasive Bladder Cancer (NMIBC) patients cancer-free long term, which has not been proven.
“Furthermore, QUILT-3.032 did not provide interpretable results on disease-free survival (DFS), and we are not aware of data that support the efficacy claims and representations that ANKTIVA can cure cancer,” the letter read.
The FDA has asked ImmunityBio to respond within 15 days, fix the misleading promotions, and warned that failure to comply could lead to legal action.
ANKTIVA, an immunotherapy to fight cancer, is approved in the U.S. for certain bladder cancer patients who no longer respond to Bacille Calmette-Guérin (BCG) treatment. The drug was recently added to Macau’s drug list and included in updated guidelines by the National Comprehensive Cancer Network as a recommended option.
ImmunityBio is also expanding ANKTIVA globally, with ongoing lung cancer trials and recent approvals in markets including Saudi Arabia.
How Did Stocktwits Users React?
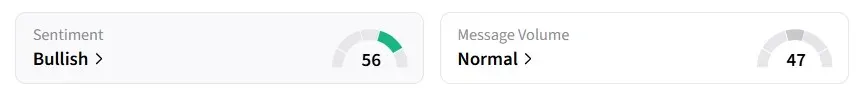
Despite the sharp intraday decline, retail sentiment for IBRX on Stocktwits flipped to ‘bullish’ from ‘bearish’ a day earlier.
One user called FDA’s warning “noise, not thesis break.”
IBRX stock recorded its biggest intraday slump since December 2024.
Another user called the stock’s decline “significant” and said the FDA letter warrants a response from management.
Year-to-date, the stock has gained more than 250%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2239579889_jpg_526b21945a.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_arthur_hayes_OG_jpg_734ff95af6.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_redwire_logo_jpg_9c82781656.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2262656307_jpg_562c79e1bd.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2227347233_jpg_00e33be418.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)