Advertisement|Remove ads.
ImmunityBio Founder Reaffirms Grip As Stock Plunges In Worst Selloff Since 2024 After Massive Rally

- Founder Patrick Soon-Shiong reaffirmed control of the company with a 66.3% stake, or 745.6 million shares.
- The ownership is spread across affiliated entities, including Nant Capital, Cambridge Equities and California Capital Equity.
- Anktiva’s expanding approvals and commercial traction continue to underpin investor interest.
Advertisement|Remove ads.
ImmunityBio, Inc. (IBRX) founder Patrick Soon-Shiong reaffirmed his grip on the cancer drugmaker in a new regulatory filing, even as shares suffered their worst day in over a year following a sharp rally driven by momentum around its immunotherapy Anktiva and a string of regulatory wins.
IBRX stock slid over 18% on Wednesday to $9.54, though it remained up nearly 18% for the week, marking its steepest single-session decline since December 2024.
Soon-Shiong Holds 66.3% Stake In ImmunityBio
A new regulatory filing on Wednesday showed that Soon-Shiong currently owns 745.6 million shares of ImmunityBio, or 66.3% of the company’s stake, based on 1.03 billion shares outstanding as of Feb. 19.
Advertisement|Remove ads.
The filing shows holdings spread across a group of affiliated entities, including Cambridge Equities, Nant Capital, California Capital Equity, and NantWorks. Cambridge reported 261.7 million shares, or 25.5% of the company, while Nant Capital reported 339.5 million shares, or 30.3%.
A key component of Nant Capital’s reported position is 93.1 million shares it has the right to acquire within 60 days through conversion of a promissory note. Soon-Shiong’s total also includes shares he can acquire within 60 days through stock options and restricted stock units.
ImmunityBio Heads Into ASCO GU 2026
The ownership update comes ahead of ImmunityBio’s participation in the ASCO Genitourinary Cancers Symposium running Feb. 26 to Feb. 28, 2026.
Advertisement|Remove ads.
The conference, themed “Patient-Centered Care: From Discovery to Delivery,” focuses on translating research advances into clinical practice and patient outcomes, with sessions spanning prostate cancer, urothelial carcinoma and kidney and testicular cancers.
Anktiva Momentum Builds Globally
The recent surge in retail focus comes amid a stream of regulatory and commercial updates around ImmunityBio’s cancer immunotherapy Anktiva.
ImmunityBio said Anktiva, in combination with BCG, is authorized for BCG-unresponsive non-muscle-invasive bladder cancer across major jurisdictions, including U.S. approval in April 2024, U.K. authorization in July 2025, and a recent conditional marketing authorization from the European Commission, along with approvals from the Saudi Food and Drug Authority.
Advertisement|Remove ads.
Saudi regulators granted accelerated approval last month for Anktiva in combination with a checkpoint inhibitor for metastatic non-small cell lung cancer.
The company reported full-year 2025 net product revenue of $113 million for Anktiva, up 700% from the previous year. Fourth-quarter net product revenue was $38.3 million, up 431% from a year earlier, while net loss attributable to common shareholders narrowed to $351.4 million from $413.6 million in 2024.
H.C. Wainwright on Monday reiterated a ‘Buy’ rating and raised its price target to $15 from $10, citing the removal of regulatory overhangs and expanding commercial reach.
Advertisement|Remove ads.
How Did Stocktwits Users React?
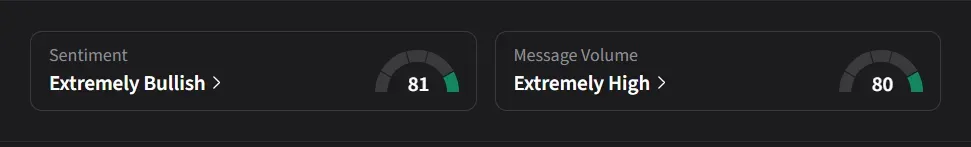
On Stocktwits, retail sentiment for IBRX has remained in the ‘extremely bullish’ territory over the past three months, driven by a 150% surge in message volumes over the past month. The stock ranks among the platform’s top three most active tickers and saw its watcher count rise by 11% over the past month.
One bullish user claimed, “momentum reset. Shaking some apples out of the tree.”
Advertisement|Remove ads.
Another user said, “Have your cake and eat it too. What a ride the last 2 weeks were. And to top it off, we now get a chance to buy it at a great discount.”
IBRX stock has skyrocketed by more than 380% year to date.
Advertisement|Remove ads.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Ark_Invest_871846bfd7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_michael_saylor_OG_4_jpg_292dd07afc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_grayscale_OG_jpg_c35dc5b1a2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_andre_francois_mckenzie_i_G_Yi_Bhd_N_Tp_E_unsplash_jpg_3d063f0b23.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1036973476_jpg_734afc6bfc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Bitcoin_and_Ethereum_2b4356b70a.webp)