Advertisement|Remove ads.
NVAX Stock Eyes Breakout Week: Novavax Claims Patients Were ‘Twice As Likely’ To Pick Its COVID Shot Over Moderna Again
- Novavax posted Q1 revenue of $139.5 million, above analysts’ estimate of $78.3 million, helped by licensing agreements.
- The drugmaker also said that patients who received Nuvaxovid were “twice as likely” to say they would choose the same vaccine again the following year.
- The company maintained its 2026 adjusted revenue outlook of $230 million to $270 million.
Shares of Novavax (NVAX) are headed for a breakout week as the vaccine maker reported solid first quarter (Q1) results and spotlighted data showing that patients were “twice as likely” to choose its COVID shot again over a rival vaccine from Moderna
NVAX stock jumped 16% on Wednesday to end at $9.36. Shares are eyeing their best week in about nine months.
Novavax Touts Lower Side Effects Vs Moderna
Novavax reported Q1 revenue of $139.5 million, beating analysts’ estimate of $78.3 million. The results were boosted by upfront and milestone payments from licensing and vaccine supply partnerships as the company shifts away from relying primarily on direct COVID vaccine sales.
The rally also came as investors focused on fresh data comparing Novavax’s protein-based COVID vaccine, Nuvaxovid, against Moderna’s mNEXSPIKE shot.
During the earnings call, Novavax highlighted findings from Sanofi’s head-to-head Phase IV Compare study, which tested Nuvaxovid directly against Moderna’s vaccine. The Maryland-based company said Nuvaxovid showed “statistically fewer side effects compared to mNEXSPIKE, across all prespecified endpoints.”
Novavax added that people who received Nuvaxovid were “twice as likely as mNEXSPIKE recipients to say they would definitely choose the same vaccine type again the following year.” The company also pointed to real-world data from the Shield study conducted with the University of Utah, saying that participants who received Nuvaxovid reported fewer side effects and “half as many hours of lost work” compared with recipients of mRNA vaccines.
The findings are notable since Novavax’s turnaround depends on its protein-based vaccine as a differentiated alternative in the crowded COVID and future combination flu-COVID market. Novavax believes the combined COVID-flu vaccine market could eventually represent a $10 billion to $12 billion opportunity.
Pfizer, Sanofi Deals Fuel NVAX Optimism
While demand for standalone COVID vaccines has weakened globally, Novavax is leaning on licensing agreements and partnerships to drive growth. The company received a $30 million upfront payment during the quarter from its Matrix-M adjuvant agreement with pfizer. Matrix-M is Novavax’s in-house ingredient that helps the body build a stronger immune response after vaccination.
“Despite whatever is happening in the macro environment, there seems to be a very strong interest,” CEO John C. Jacobs said during the conference call. The company said that its partners are currently exploring over 30 infectious disease and cancer indications using Matrix-M.
“Our partnerships with Sanofi and Pfizer have the potential on their own, to generate billions in revenue from milestone and royalty opportunities,” Jacobs said. Sanofi is developing combination flu-COVID vaccines using Nuvaxovid under a deal worth at least $1.2 billion.
NVAX Reaffirms 2026 Revenue Outlook
Novavax maintained its 2026 adjusted revenue forecast in the range of $230 million and $270 million, excluding royalties and sales from the Sanofi agreement. The company also reiterated that it expects its existing cash position and partner reimbursements to fund operations into 2028 even without additional milestones or royalty revenue. Novavax posted a Q1 net loss of just $9 million and continues targeting non-GAAP profitability by 2028.
After the earnings report and updated vaccine data, TD Cowen raised its price target on Novavax to $9 from $8, still implying a 4% downside from current levels, while maintaining a ‘Hold’ rating. The brokerage cited the Sanofi-led head-to-head study and growing evidence that Nuvaxovid may offer a differentiated tolerability profile compared with other mRNA vaccines.
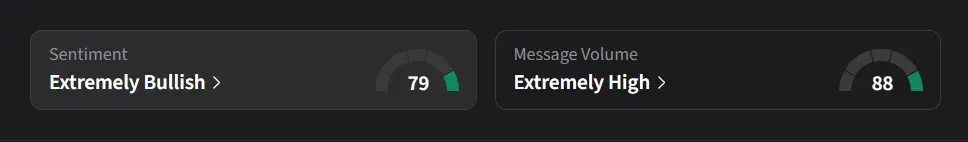
How Do Retail Traders Feel About NVAX?
On Stocktwits, retail sentiment for NVAX was ‘extremely bullish’ amid over a 430% surge in 24-hour message volumes.
One user said, “not chasing but definitely watching this closely”
Another user said, “Lean organization, massive $100 billion market. No rush. Im not expecting to be rich tomorrow but I do expect it to happen in a year”
NVAX stock has risen 54% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: Nio Stock Jumps In HK: ES9 Launch Buzz Drowns Out Fresh UBS Sell-Down











/filters:format(webp)https://news.stocktwits-cdn.com/large_doordash_resized_jpg_e2e74f3ef9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2232147543_jpg_a7d6168b5c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_supermicro_resized_jpg_95d12828d5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_novaxovid_novavax_resized_jpg_3a4b0527ae.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_nio_stock_jpg_770e12377f.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2252956558_jpg_2dc0e5e537.webp)