Advertisement|Remove ads.
RIGL Stock Jumps 15%, Short Interest Surges To Record High – Why Is Retail Eyeing A Further 40% Upside?

- Rigel will develop, manufacture, and commercialize Vepdegestrant, which will be sold under the brand name Veppanu.
- Vepannu, developed by Arvinas and Pfizer, received FDA approval earlier this month.
- Arvinas and Pfizer will receive $70 million upfront and an additional $15 million upon completion of certain transition activities.
Shares of Rigel Pharmaceuticals, Inc (RIGL) surged more than 15% on Tuesday after investors welcomed a potential growth opportunity tied to the company’s newly signed global licensing deal with Arvinas (ARVS) and Pfizer (PFE) for a breast cancer treatment.
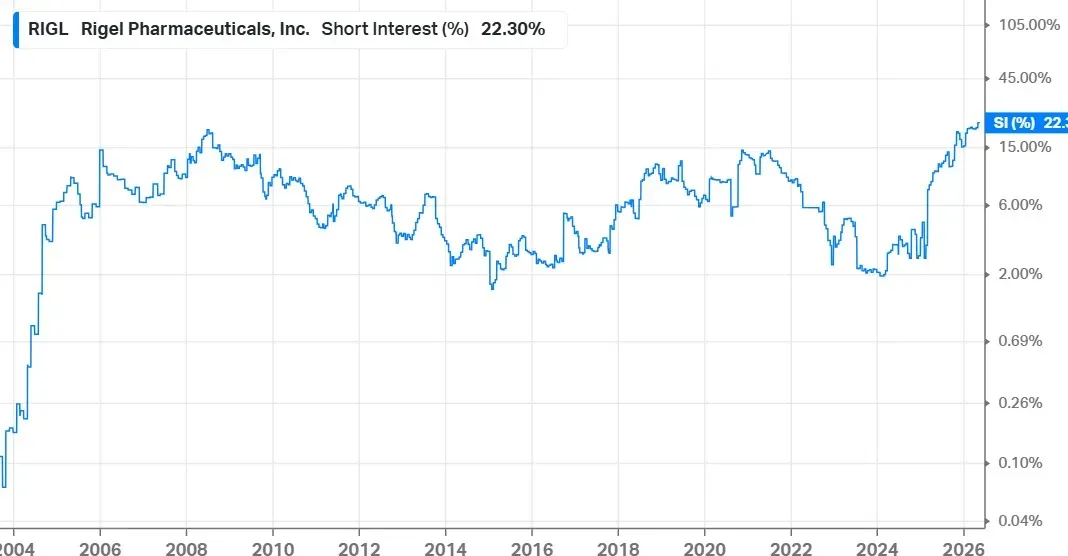
According to Koyfin data, short interest in RIGL climbed to 22.3% of the float as of May 12, up sharply from 15.5% at the beginning of the year, marking its highest level ever. The company has roughly 18.5 million outstanding shares.
Source: Koyfin
Veppanu To Drive RIGL’s Revenue Growth
Under the agreement, Rigel will develop, manufacture, and commercialize Vepdegestrant, which will be sold under the brand name Veppanu. The drug is the only FDA-approved oral PROteolysis TArgeting Chimera (PROTAC) therapy, a new class of medicines designed to target and break down disease-causing proteins.
Arvinas and Pfizer will receive $70 million upfront and an additional $15 million upon completion of certain transition activities. The companies could also receive up to $320 million in future milestone payments.
Rigel said Veppanu will become its fourth commercial product and a key driver of future revenue growth as it expands its cancer treatment portfolio.
“Veppanu is expected to contribute strong revenue growth in our expanding commercial portfolio, and we believe it has the potential to become a meaningful driver of long-term growth for Rigel,” said Raul Rodriguez, Rigel’s president and CEO.
Vepannu’s Phase 3 Data Shows 43% Fall In Disease Progression Risk
The drug, developed by Arvinas and Pfizer, received FDA approval earlier this month. The regulatory clearance was supported by a Phase 3 study showing that Veppanu reduced the risk of disease progression or death by 43% compared with the comparator cancer drug, Fulvestrant. Patients treated with Veppanu had a median progression-free survival of five months, compared with 2.1 months for Fulvestrant.
Following the FDA approval, Arvinas announced plans to partner with a third party to support the drug’s commercial rollout. Veppanu received Fast Track designation from the FDA in 2024, followed by the agency’s acceptance of its New Drug Application (NDA) for review last August.
RIGL Bulls Eyes $50
Retail sentiment for RIGL shares on Stocktwits turned ‘extremely bullish’ from ‘bullish’ a day earlier, amid ‘extremely high’ message volumes.
One user expects the stock to climb to $50, a 40% upside from current levels, stating Rigel is no longer “just a company managing a future patent cliff” but is a “growing commercial oncology company.”
Another user called the deal a “potential game-changer.”
The stock has gained more than 14% so far in 2026.
Read also: NVDA Stock Has Gained Nearly 16% In A Month – But Wall Street Sees Further Upside
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2257538219_jpg_1a53bb8d9a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246580703_jpg_9700e1e7e8.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201667454_jpg_2f4f6cc4ac.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269641966_jpg_a7691989ef.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_Stock_market_Image_public_domain_declining_wc_96197f57d2.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2202932376_jpg_32367a92e5.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Chinmay_Rautmare_Photo_jpeg_96dd37efd0.webp)