Advertisement|Remove ads.
Why Did KOD Stock Surge To A Four-Year High Today?
- Zenkuda achieved at least a two-step improvement in DRSS score compared with just 3.3% in the control group.
- The eye drug also reduced the risk of sight-threatening complications by 85% through Week 48.
- Zenkuda delivered consistent results regardless of patients using GLP-1.
Shares of Kodiak Sciences (KOD) jumped nearly 60% on Thursday, after the pharmaceutical firm reported positive Phase 3 results for its eye drug Zenkuda to treat patients with diabetic retinopathy.
KOD shares are trading at their highest levels since February 2022.
Study Meets Primary Endpoint
The study met its primary endpoint as a total of 62.5% of patients treated with Zenkuda achieved at least a two-step improvement in DRSS score, a measure of severity, by week 48, compared with just 3.3% in the control group. The drug also reduced the risk of sight-threatening complications by 85% through Week 48.
Zenkuda delivered consistent benefits regardless of GLP-1 use, with 60% of patients on GLP-1 therapies achieving a two-step DRSS improvement by Week 48, compared to 64.3% among those not using GLP-1 drugs. The treatment was well tolerated, with no cases of intraocular swelling and low rates of side effects.
“We now have a multi-indication BLA-ready profile for Zenkuda, and we intend to accelerate our BLA submission plans,” said Victor Perlroth, CEO of Kodiak Sciences.
How Did Retail Investors React?
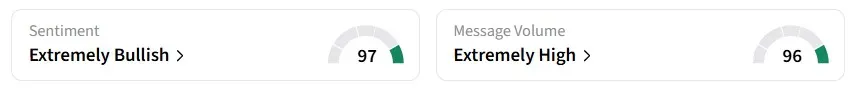
Retail sentiment on Stocktwits remained in the 'extremely bullish' zone over the past 24 hours, amid 'extremely high' message volumes.
One user called the results, "very good."
Another user highlighted the treatment's potential commercial value.
KSI-101 Trial Update
Last month, Kodiak Sciences presented new clinical data from Phase 1b APEX study results for the treatment of patients with macular edema linked to inflammation.
The company’s drug KSI-101 targets both IL-6 and VEGF and showed meaningful vision and retinal improvements in early testing. The treatment was also well tolerated across patient groups.
Based on the results, the company has advanced two dose levels into late-stage trials. Phase 3 studies are now underway to further evaluate the therapy.
Year-to-date, KODK shares have gained around 30%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1233265488_jpg_60af97169a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275851163_jpg_b115c861be.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2237640344_jpg_bc97a7240c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2227347116_jpg_6a738ad597.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201127733_jpg_293396ee32.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269243885_jpg_4cc426aadd.webp)