Advertisement|Remove ads.
Why Did NGNE Stock Surge 25% In Pre-Market Today?
- The FDA decision was based on interim Phase 1/2 trial data showing meaningful and lasting improvements across multiple Rett syndrome domains.
- The company plans to share additional interim clinical data in mid-2026.
- Canaccord initiated coverage of Neurogene with a ‘Buy’ rating and a price target of $200, according to The Fly.
Advertisement|Remove ads.
Shares of Neurogene Inc. (NGNE) jumped 25% in premarket trading on Friday after a bullish analyst initiation and news that the U.S. Food and Drug Administration granted a key designation for its investigational gene therapy targeting Rett syndrome.
If the pre-market levels hold, NGNE shares would decisively break above their 200-day moving average (200-DMA) for the first time since January 5, and trade at their highest levels since Nov. 14, 2025.
Advertisement|Remove ads.
Source: TradingView
Canaccord analyst Whitney Ijem initiated coverage of Neurogene with a ‘Buy’ rating and a price target of $200, according to The Fly. This represents a 916% upside to Thursday’s closing price of $19.68.
FDA Grants Breakthrough Therapy Designation
After market hours on Thursday, Neurogene announced that the U.S. FDA granted Breakthrough Therapy designation to its experimental gene therapy, NGN-401, for the treatment of Rett syndrome. According to the FDA, the designation is intended to speed up the development and review of drugs for serious conditions when early clinical evidence suggests the therapy could offer significant improvement over existing treatments on meaningful clinical endpoints.
Advertisement|Remove ads.
The decision was based on interim Phase 1/2 trial data showing meaningful and lasting improvements across multiple Rett syndrome domains. The FDA evaluated safety and efficacy findings, including detailed patient-level data and supporting video evidence, using results gathered through Oct. 30, 2025.
NGN-401 is designed as a one-time treatment that delivers the full-length human MECP2 gene directly to the brain and nervous system. The therapy is currently being studied in the Embolden registrational trial, with dosing expected to be completed in the second quarter of 2026. The company also plans to share additional interim clinical data in mid-2026.
How Did Stocktwits Users React?
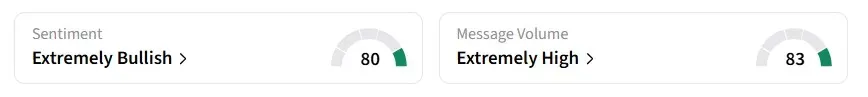
Retail sentiment on Stocktwits shifted to ‘extremely bullish’ from ‘bullish’ a day earlier, amid ‘extremely high’ message volumes.
Advertisement|Remove ads.
A bullish user expects the stock to climb to $35.
Advertisement|Remove ads.
Year-to-date, the stock has climbed 13%.
Also See: AAOI Stock On Track To Open At Over 8-Year Highs: B. Riley Upgrade, Retail Optimism Drive Rally
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Advertisement|Remove ads.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_AOL_jpg_cf55f24cd0.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Tom_Lee_c6db3cdfe4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_RTX_corp_resized_b411f67ef2.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Chinmay_Rautmare_Photo_jpeg_96dd37efd0.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2213582854_jpg_830e44a354.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_826431872_jpg_683327871a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1401171512_7_jpg_ff7adea960.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_6979_jpg_a2a1032fdc.webp)