Advertisement|Remove ads.
Why Did ALDX Stock Tank 70% Today?

- FDA said the application has failed to demonstrate efficacy in adequate and well controlled studies in the treatment of signs and symptoms of dry eye disease.
- "Inconsistency of study results raises serious concerns about the reliability and meaningfulness of the positive findings,” the FDA said.
- Aldeyra does not plan to conduct additional trials and will instead request a Type A meeting with the FDA.
Advertisement|Remove ads.
Shares of Aldeyra Therapeutics Inc. (ALDX) plummeted nearly 70% on Tuesday, after the U.S. Food and Drug Administration issued a complete response letter stating insufficient and inconsistent clinical data for the company’s dry eye treatment.
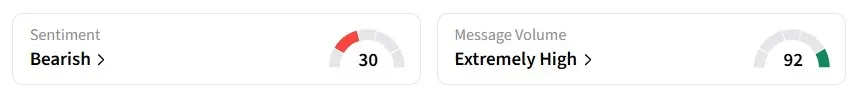
The latest update generated a significant buzz on Stocktwits, with retail sentiment for ALDX on flipping to ‘bearish’ from ‘bullish’ a day earlier. Message volumes on the platform were ‘extremely high.’
Advertisement|Remove ads.
FDA Says Inconsistencies Raises Serious Concerns
Responding to the company’s application for reproxalap, the FDA cited “a lack of substantial evidence consisting of adequate and well-controlled investigations … that the drug product will have the effect it purports or is represented to have under the conditions of use prescribed, recommended, or suggested in its proposed labelling.”
The letter also read “the application has failed to demonstrate efficacy in adequate and well controlled studies in the treatment of signs and symptoms of dry eye disease.” The FDA stated “inconsistency of study results raises serious concerns about the reliability and meaningfulness of the positive findings” and that the “totality of evidence from the completed clinical trials does not support the effectiveness of the product.”
What Did The FDA Recommend?
The FDA advised analyzing why certain trials failed and identifying specific patient groups or conditions where reproxalap may be effective. The administration did not recommend conducting additional trials or request submission of additional evidence.
Advertisement|Remove ads.
As per prior NDA reviews of the drug, no safety or manufacturing concerns were identified, the company added.
Aldeyra said that it does not plan to conduct additional trials and will instead request a Type A meeting with the FDA to determine the steps needed for approval.
What Did Stocktwits Users Say?
One user said the FDA’s response essentially means “back to square one” for the company.
Advertisement|Remove ads.
Another user said the field trial is what “did them in.”
Advertisement|Remove ads.
Year-to-date the stock has declined around 75%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_arthur_hayes_OG_jpg_734ff95af6.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_strategy_logo_OG_jpg_fa4e1a7d04.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1376765653_jpg_d56a66290e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275170769_jpg_e811c3ae51.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spacex_jpg_6eb3abcb72.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2271060260_jpg_b0369496dd.webp)