Advertisement|Remove ads.
HIMS Stock On The Mend? New Testosterone Push Builds As GLP-1 Gold Rush Gets Crowded

- The company launched Testosterone Rx+, a once-daily pill designed to help the body naturally increase testosterone levels.
- The rollout follows a recent FDA order supporting expanded testosterone therapy use for men with low libido from idiopathic hypogonadism.
- The testosterone push comes as the FDA proposes restricting compounding of GLP-1 ingredients such as Semaglutide and Tirzepatide.
Shares of Hims & Hers Health, Inc. (HIMS) snapped a three-day losing streak on Thursday after the telehealth firm expanded its testosterone-care offering, creating a new growth pillar as competition intensifies across the crowded GLP-1 weight-loss market.
HIMS stock ended over 3% higher at $27.17 on Thursday, snapping three straight sessions of losses. Shares also logged their second consecutive month of gains, up 31% in April.
Hims Turns Up Its Testosterone Push
The company on Thursday introduced Testosterone Rx+, a once-daily pill that naturally increases testosterone levels, as part of its broader push into men’s hormone therapies following a recent regulatory push supporting the use of testosterone therapy.
Recently, the U.S. Food and Drug Administration (FDA) encouraged sponsors of approved testosterone replacement therapies to explore a potential new indication for treating low libido from idiopathic hypogonadism.
Hims began building its testosterone platform in September 2025, allowing providers to prescribe Enclomiphene-based treatments that help the body produce more of its own testosterone, while preserving fertility. The company also introduced combination therapy with Tadalafil, a medication used to treat erectile dysfunction, for men with low testosterone.
Testosterone Rx+ builds on the same approach by combining Enclomiphene with zinc, vitamins B6 and B12, and L-arginine, an amino acid for better blood flow, in a single pill. According to company data, 95% of customers saw increases in testosterone levels, with 80% reaching optimal levels within 60 days.
The program also includes symptom-screening tools, at-home lab testing kits, and virtual care through the platform.
GLP-1 Crackdown Lifts Testosterone Bet
The testosterone expansion comes as regulators tighten oversight around compounded obesity medicines that have previously benefited telehealth firms.
The FDA recently proposed excluding key GLP-1 ingredients, such as Semaglutide and Tirzepatide, from its 503B bulk list, which determines when outsourcing facilities can compound medicines from raw ingredients. The proposal could limit the large-scale availability of compounded versions and increase reliance on branded therapies, underscoring the importance of additional revenue engines, such as testosterone care, within Hims’ portfolio.
Earlier this year, Novo Nordisk filed a patent infringement lawsuit related to compounded semaglutide offerings before reaching an agreement with Hims to provide access to FDA-approved versions of Wegovy and Ozempic through the platform.
GLP-1 Rivals Crowd The Field
Competition across the weight-loss treatment ecosystem continues to intensify as Novo Nordisk and Eli Lilly scale blockbuster GLP-1 therapies, while large tech players such as Amazon also enter the weight-loss space.
Firms such as Structure Therapeutics are advancing an oral GLP-1 candidate that showed more than 16% weight loss in mid-stage trials, while Altimmune is progressing its Pemvidutide program toward Phase 3 development after aligning trial plans with the FDA.
Meanwhile, upcoming FDA reviews of peptide substances for possible inclusion on the federal 503A bulks list could create another lifeline for Hims to expand peptide-based treatment offerings on its platform.
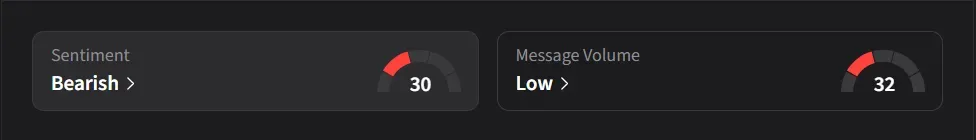
How Do Retail Traders Feel About HIMS?
On Stocktwits, retail sentiment was ‘bearish’ amid ‘low’ message volume.
One user said, “Peptides are coming in July. See you at $70 in September!”
Another user said, Good to see the FDA helping the cause by making drug development a business that should be protected.”
HIMS stock has declined 18% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2236688965_jpg_b00d009983.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_arthur_hayes_2026_OG_jpg_9f3524f8b1.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2261265006_jpg_cd8b462617.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2256966922_jpg_81b5386f83.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_zoom_video_logo_resized_15c98df8d1.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_viasat_resized_50c4271397.jpg)