Advertisement|Remove ads.
IBRX Stock Still A Buy After Worst Day In Over A Year? Why This Analyst Is Shrugging Off FDA Warning Letter

- BTIG maintained its 'Buy' rating and $13 price target, implying about 75% upside, and said the warning relates to promotion rather than safety or efficacy risks.
- The brokerage highlighted second-half 2026 data as a key upcoming catalyst.
- ImmunityBio continues advancing Anktiva combination studies, including a Phase 1 pancreatic cancer trial.
Shares of ImmunityBio, Inc. (IBRX) jumped over 2% in overnight trading on Wednesday after an analyst said the recent FDA warning letter tied to Anktiva's promotional claims is likely resolvable, and that the pullback represents a potential entry opportunity ahead of key data this year.
IBRX stock plunged over 21% on Tuesday to end at $7.42, logging its worst session since December 2024.
BTIG Sees Buying Opportunity After Selloff
BTIG said the warning letter relates to promotional messaging rather than product safety or efficacy, and does not expect legal action or product withdrawal, citing precedent from the FDA’s Office of Prescription Drug Promotion and viewing the issue as likely resolvable.
The brokerage also called the pullback a potential entry opportunity ahead of the second-half 2026 data in bladder cancer patients who have not yet received BCG treatment, which remains a key upcoming catalyst.
BTIG maintained its ‘Buy’ rating and $13 price target, implying a 75% upside to current levels.
FDA Flags Promotional Claims On Anktiva
The warning letter cited statements made in a television advertisement and during a January appearance by executive chairman Patrick Soon-Shiong on The Sean Spicer Show, where he suggested Anktiva could treat “all cancers.”
The FDA said the communications promoted uses beyond the therapy’s approved indication and lacked adequate directions for those broader claims. The agency also objected to statements describing Anktiva as a cancer vaccine that “can treat the tumor and it doesn’t come back.”
The regulatory pushback follows a broader campaign by Soon-Shiong urging regulators to consider expanded approval pathways for Anktiva after the FDA earlier declined to review a supplemental application seeking broader use. ImmunityBio resubmitted its request earlier this month to expand the drug’s label to papillary bladder cancer, which could increase its commercial opportunity if approved.
Anktiva Advances Across Multiple Cancer Studies
ImmunityBio continues advancing Anktiva across multiple tumor settings, including a Phase 1 pancreatic cancer study evaluating the therapy in combination with the investigational agents Sotevtamab and Zabadinostat, alongside the chemotherapy drugs gemcitabine and Bristol Myers Squibb’s Abraxane.
The company is positioning Anktiva as a platform immunotherapy designed to work with vaccines, checkpoint inhibitors and chemotherapy across cancers, supported by early combination-study data showing increases in absolute lymphocyte counts.
Meanwhile, Anktiva was recently added to Macau’s approved bladder cancer drug list and included by the National Comprehensive Cancer Network (NCCN) with standard BCG treatment as an “other recommended” option for papillary bladder cancer after failure of standard BCG therapy, expanding its role beyond carcinoma in situ under treatment guidelines.
How Did Stocktwits Users React?
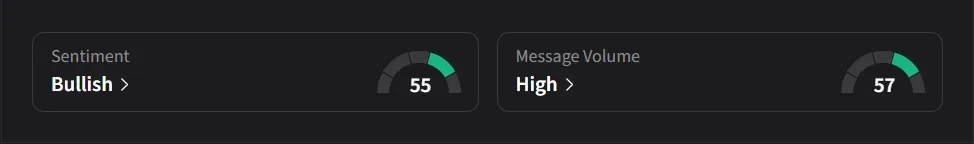
On Stocktwits, retail sentiment for IBRX jumped to ‘bullish’ from ‘bearish’ levels over the past day amid over a 200% surge in 24-hour message volume.
One user said, “The FDA warning letter sounds scary in headlines but it’s a promotional compliance issue for $IBRX , not a safety or efficacy problem with ANKTIVA itself. They can fix their marketing materials, submit a remediation plan, and move on.”
Another user hopes to see a “meaningful rebound on the upward trend” on Wednesday.
IBRX stock has surged over 160% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)