Advertisement|Remove ads.
IBRX Stock Can't Shake FDA Delay Overhang: Founder Touts Saudi And Greece Progress With Anktiva As Bright Spots

- Soon-Shiong highlighted that ImmunityBio’s Quilt 3005 immunotherapy is already approved by the Saudi FDA.
- Investors are closely watching the FDA’s ongoing review of ImmunityBio’s resubmitted BLA for Anktiva plus BCG in bladder cancer.
- The filing is backed by Quilt 3.032 data, which showed that 58.2% of patients remained cancer-free after 12 months.
Shares of ImmunityBio, Inc. (IBRX) slid over 1% in overnight trading heading into Monday amid lingering review delays from the U.S. Food and Drug Administration (FDA) for the company’s cancer immunotherapy, with founder Patrick Soon-Shiong pointing to Saudi Arabia’s approval and ongoing talks with Greece as silver linings.
IBRX stock snapped a two-week losing streak last week, surging 20% for its strongest weekly performance since February.
IBRX Greece Meetings Fuel Expansion Buzz
Patrick Soon-Shiong said on X that the current cancer treatment methods relying on chemotherapy, radiation and checkpoint inhibitors need to evolve toward immunotherapy after citing a New York Times report, which showed that nearly half of advanced lung cancer patients do not receive treatment and about 40% die within three months of diagnosis.
“Chemo. Radiation then checkpoints, then even more chemo!!” Soon-Shiong said. “When do we learn that this can change with immunotherapy?” He then pointed to ImmunityBio’s Quilt 3005 program, adding: “Quilt 3005 now approved by Saudi FDA.”
The remarks drew investor attention because ImmunityBio’s immunotherapy platform has already gained approval in Saudi Arabia for certain lung cancer patients, while the company continues navigating additional FDA reviews and filings in the U.S.
Soon-Shiong separately posted on X on Sunday about meetings in Greece with Prime Minister Kyriakos Mitsotakis, Health Minister Adonis Georgiadis and Ecumenical Patriarch Bartholomew I. “N.K. cells are God’s gift to humanity and Greece has embraced this for their people,” Soon-Shiong said. “Global expansion continues.”
Anktiva FDA Review In Focus
Another major focus for investors is the FDA’s ongoing review of ImmunityBio’s resubmitted supplemental biologics license application (BLA) for Anktiva, plus standard BCG treatment, in patients with non-muscle-invasive bladder cancer with papillary tumors. The FDA’s standard 60-day filing review window ends anytime now.
The filing is backed by long-term data from the Quilt 3.032 study, which ImmunityBio said showed that 58.2% of patients remained cancer-free after 12 months, while more than 80% avoided bladder-removal surgery over three years.
IBRX Investors Watch FDA Shakeup
The FDA backdrop has become even more closely watched after recent turmoil surrounding Commissioner Marty Makary. The Wall Street Journal reported on Friday that U.S. President Donald Trump had signed off on a plan to potentially remove Makary after disputes stemming from vaping approvals, abortion-pill reviews and broader drug policy disagreements.
However, Trump later downplayed the report, saying that he knew “nothing about it.” Makary has faced criticism from biotechnology executives, rare-disease advocates and patient groups over FDA review standards and approval decisions involving experimental therapies.
The uncertainty matters for biotech companies like ImmunityBio since former Center for Biologics Evaluation and Research (CBER) official Vinay Prasad faced criticism for rejecting or delaying several experimental cancer and rare-disease therapies, while raising approval standards and creating hurdles for accelerated pathways.
ImmunityBio Pushes Overseas Expansion
ImmunityBio has continued to expand globally even as U.S. regulatory scrutiny remains in the spotlight. Earlier this year, the company launched Anktiva in Saudi Arabia for certain patients with bladder and lung cancer through partnerships with Biopharma and Cigalah Healthcare as part of a broader Middle East and North Africa expansion.
Anktiva, which was first approved by the U.S. Food and Drug Administration in 2024 for a form of bladder cancer, has since gained approvals in the United Kingdom, European Union and Macau. The European Commission later expanded authorization across 33 countries, while the National Comprehensive Cancer Network (NCCN) added the therapy as an “other recommended” option for papillary bladder cancer.
The company also recently faced pressure after the FDA issued a warning letter for promotional claims suggesting broader cancer uses beyond Anktiva’s currently approved bladder cancer indication.
How Do Retail Traders Feel About IBRX?
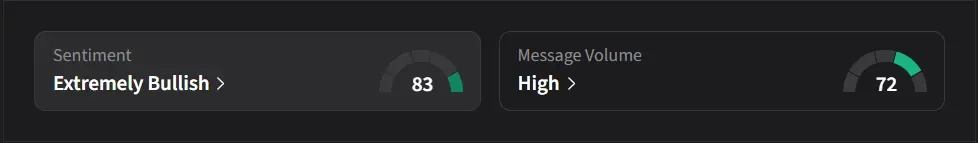
On Stocktwits, retail sentiment for IBRX was ‘extremely bullish’ amid ‘high’ message volume.
One user said, “Each country Patrick locks up the more pressure is on our FDA and Trump. Horrifying look the world is progressing with an American company while America does nothing. This cannot stand. No way.”
Another user said, “I got a feeling our good Dr. Pat will have a string of PRs coming from various countries.”
IBRX stock has surged 307% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2249184989_jpg_2f36d82a30.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_950698324_jpg_d6201b2562.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_951165788_jpg_24c6559b2b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1368998806_2_jpg_e532306557.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1163013566_jpg_dc3a81571d.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2229273884_jpg_b009b52b81.webp)