Advertisement|Remove ads.
IBRX Stock Extends Rally: Founder Flags Fresh Bull Case For Anktiva On Breast Cancer Immune Signal

- Founder Patrick Soon-Shiong highlighted new breast cancer immunotherapy research that supports Anktiva’s scientific rationale.
- A Phase 2 study is already evaluating Anktiva in persistent post-COVID symptoms.
- Anktiva, approved in 2024 with BCG for certain bladder-cancer patients, is now being studied across multiple cancers, including lung and pancreatic indications.
Shares of ImmunityBio, Inc. (IBRX) extended their rally on Monday after founder Patrick Soon-Shiong cited emerging breast-cancer research linking low immune-cell levels to poorer outcomes, reinforcing the scientific case for Anktiva, its flagship immunotherapy.
IBRX stock jumped over 6% on Monday, logging its second straight session of gains.
Immune-Cell Findings Support Anktiva Case
Soon-Shiong called the findings on X as part of a “drumbeat of evidence” showing that low levels of key immune cells are linked to weaker treatment response and a higher risk of breast cancer recurrence.
Over the weekend, he said patients in remission with early-stage breast cancer who develop lymphopenia, which is reduced levels of T cells and natural killer (NK) cells, face “an INCREASE OF OVER 2 TIMES THE RISK of distant recurrence.” He added that measuring absolute lymphocyte count is “an easy test” that can help identify patients at higher risk.
The immune-cell findings are notable because Anktiva can boost natural killer (NK) cells and CD8 T cells, which are key components of the immune system that help the body identify and destroy cancer cells.
Soon-Shiong earlier said clinicians at the University of California, Los Angeles and Mount Sinai Health System observed links between post-COVID immune-cell declines and rising rates of metastatic disease and cancer recurrence in patients who had been in remission. He added that the observations are now “being validated” by emerging data presented at the ongoing American Association for Cancer Research (AACR) Annual Meeting. “The relationship of low NK and low T cells to breast cancer (and frankly all cancers) is so clear,” he said.
IBRX Studies Anktiva In Post-COVID Recovery
Additionally, Soon-Shiong said over the weekend that a patient is currently being treated under an FDA-authorized single-patient investigational pathway to evaluate whether immune dysfunction linked to post-COVID lymphopenia can be reversed.
“This is a clinical study and not an approved indication, but we are finding what is known as T-cell exhaustion,” he said, adding that additional clinical trials are planned to explore whether restoring immune-cell balance could reduce recurrence risk.
ImmunityBio is already evaluating Anktiva in a Phase 2 study in patients with persistent post-COVID symptoms to test whether correcting the immune-cell imbalance may address the underlying immune dysfunction.
IBRX Expands Anktiva Beyond Bladder Cancer
Anktiva received approval in 2024 with standard BCG for certain bladder-cancer patients who no longer respond to treatment and is now being studied in non-small cell lung cancer, pancreatic cancer, colorectal cancer, glioblastoma, and immune-related conditions, including severe pneumonia, sepsis, long COVID, and acute respiratory distress syndrome.
The therapy has also secured conditional marketing authorization from the European Commission covering the European Union as well as Iceland, Liechtenstein and Norway.
How Do Retail Traders Feel About IBRX?
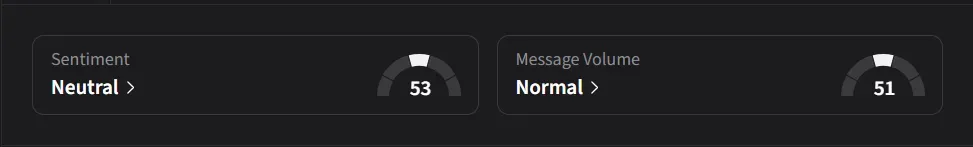
On Stocktwits, retail sentiment for IBRX was ‘neutral’ amid a 286% surge in 24-hour message volumes.
One user said, “I work with a lot of long COVID patients. I could see this indication being worth so much $ given 17mil adults in the US live with long COVID right now. I know it's years til approval, but I'm looking for a long term hold.”
Another user said, “#anktiva will resolve Lymphopenia for patients with Triple Negative Breast Cancer significantly improving their outcomes and survivability.”
IBRX stock has surged over 230% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2249184989_jpg_2f36d82a30.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_950698324_jpg_d6201b2562.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_951165788_jpg_24c6559b2b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1368998806_2_jpg_e532306557.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1163013566_jpg_dc3a81571d.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2229273884_jpg_b009b52b81.webp)