Advertisement|Remove ads.
IBRX Founder Backs Trump’s Psychedelic Push In ‘Transforming’ Health Policy — Flags New Anktiva Breast Cancer Risk Data

- The founder said Trump “can make huge advances in transforming the health of Americans” following a White House push to accelerate psychedelic-therapy research.
- He flagged evidence showing over 2x higher distant breast cancer recurrence risk in remission patients with lymphopenia, or reduced levels of key immune cells.
- Further trials are planned to test whether post-COVID immune dysfunction associated with T-cell exhaustion can be reversed with Anktiva.
Shares of ImmunityBio, Inc. (IBRX) drew fresh attention over the weekend after founder Patrick Soon-Shiong backed U.S. President Donald Trump’s psychedelic-policy push and flagged new data linking low immune-cell levels to a higher risk of breast-cancer recurrence, spotlighting Anktiva.
IBRX stock snapped a three-week losing streak to end last week up 6%.
IBRX Founder Backs Trump Psychedelic Push
Soon-Shiong responded over the weekend to a White House push to accelerate research into psychedelic therapies, including ibogaine, saying on X that the president “can make huge advances in transforming the health of Americans.”
The remarks followed an executive order directing federal support toward state-level programs advancing psychedelic treatments for serious mental illness. The development, alongside a post on X from FDA Commissioner Marty Makary that the agency is “moving proactively to advance potential therapies to address America’s mental health crisis,” a post Soon-Shiong also shared.
Podcast host Joe Rogan said during the ceremony he had earlier texted Trump about ibogaine’s addiction-treatment potential and recalled the president replying, “Sounds great. Do you want FDA approval? Let’s do it.”
New Breast Cancer Risk Data
In a separate weekend update, Soon-Shiong pointed to new findings linking lymphopenia, or reduced levels of key immune cells, to higher recurrence risk in patients with early-stage breast cancer in remission.
“News will break tomorrow,” he said. “Evidence of an INCREASE OF OVER 2 TIMES THE RISK of distant recurrence of breast cancer (in patients in remission) with lymphopenia (low TCells and low NK cells).” He added that absolute lymphocyte count “matters in patients in remission with early stage (1 to 3) breast cancer” and called it an “easy test to monitor lymphopenia and to address this increased risk.”
Soon-Shiong later cited independent research presented by UCLA breast cancer oncologist Aditya Barda at the American Association for Cancer Research (AACR) meeting, saying that the data showed that “regardless of the stage of BREAST CANCER, that post Covid lymphopenia results in poorer prognosis, poorer response to treatment and even 2x higher recurrence when patients are in remission.”
“The relationship of low NK and low T cells to breast cancer (and frankly all cancers) is so clear,” he added, calling the findings “very compelling.”
Trials Planned To Reverse Post-COVID Immune Dysfunction
Additionally, Soon-Shiong responded to questions about how clinicians may address post-COVID lymphopenia, saying a patient is currently being treated under an FDA-authorized single-patient investigational new drug pathway.
“This is a clinical study and not an approved indication, but we are finding what is known as T cell exhaustion,” he said, adding that more clinical trials are planned to explore whether the condition can be reversed.
The immune-cell findings are notable in the context of Anktiva, an IL-15-based immunotherapy that boosts natural killer cells and CD8 T cells, two immune cell types that help the body detect and destroy virus-infected and cancer cells.
Soon-Shiong had earlier said clinicians at the University of California, Los Angeles and the Mount Sinai Health System were observing links between lymphopenia after COVID and a rising incidence of metastatic disease and cancer recurrence in patients who had previously been in remission. He added that those concerns were “being validated” by emerging data presented at the AACR summit.
ImmunityBio is already evaluating Anktiva in a Phase 2 study in patients with persistent post-COVID symptoms to test whether restoring immune-cell balance may correct underlying immune dysfunction.
Founder Defends Anktiva As Policy Momentum Builds
The latest updates follow recent regulatory scrutiny of Anktiva after the FDA issued a warning letter citing promotional language that referenced uses beyond the therapy’s approved indication. Soon-Shiong pushed back publicly, saying a television advertisement cited by regulators “never aired at all.”
Anktiva, approved in 2024 with standard BCG for certain bladder-cancer patients who no longer respond to standard therapy, is now being studied across multiple tumor types and severe inflammatory conditions. The therapy has also expanded internationally after conditional marketing authorization from the European Commission across the European Union, plus Iceland, Liechtenstein and Norway.
Soon-Shiong has previously said that talks with Trump and Saudi Crown Prince Mohammed bin Salman helped accelerate regulatory progress and the global rollout of the company’s Bioshield platform, including steps supporting lung cancer approvals and regional deployment across the Middle East and North Africa.
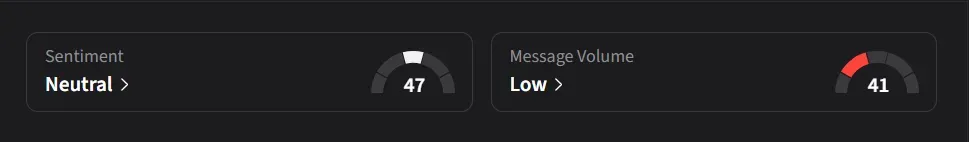
How Do Retail Traders Feel About IBRX?
On Stocktwits, retail sentiment for IBRX has improved to ‘neutral’ from ‘bearish’ levels over the past week amid a 70% jump in message volumes during the same period.
One user said, “sold a good bit Friday at 7.84 with the intention to buy my khc stock back but now I’m buying a ton of Ibrx tommorow. Don’t be a panican! A mountain of money is waiting to get in here.”
Another user expressed optimism for Anktiva’s broader approval in additional approvals saying, “zero doubt this is coming before the midterms. Trump will have the heyday of curing cancer during his term.”
IBRX stock has jumped 212% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: ASTS Stock Slumps Overnight: BlueBird-7 Orbit Failure Forces Satellite Exit From Network Buildout

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1233265488_jpg_60af97169a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275851163_jpg_b115c861be.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2237640344_jpg_bc97a7240c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2227347116_jpg_6a738ad597.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201127733_jpg_293396ee32.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269243885_jpg_4cc426aadd.webp)