Advertisement|Remove ads.
IBRX Stock Extends Rally To Day 3: Founder Drops ‘Prelude’ To 2025 Earnings, Slams Bristol Myers, Amgen Cancer Playbook

- Founder and executive chairman Patrick Soon-Shiong called a rare public speech his “prelude” to Tuesday’s earnings update.
- The company is set to report FY25 results on Tuesday, with estimates pointing to revenue of $113.3 million versus $14.8 million in 2024 and narrowing losses.
- Anktiva generated $113 million in net product revenue in 2025, up 700% year over year, while Q4 revenue climbed 431% to $38.3 million.
Shares of ImmunityBio, Inc. (IBRX) extended their three-day rally on Monday, with founder and executive chairman Patrick Soon-Shiong adding fuel to the stock by calling a rare public speech a “prelude” to its earnings update due Tuesday.
IBRX stock rose nearly 7% on Monday to end at $10.44 and slipped slightly in extended trading.
IL-15 Spotlighted As Anktiva’s Core Engine
Soon-Shiong said on X that his remarks from last week, delivered at an event marking the 55th anniversary of President Richard Nixon’s “war on cancer,” were a “prelude” to ImmunityBio’s upcoming earnings.
He criticized decades-old cancer treatment strategies, arguing that many chemotherapy drugs were developed without fully accounting for the immune system. He said high-dose chemo and radiation can significantly reduce vital immune cells like NK cells and T cells, creating a condition known as lymphopenia.
He called the absolute lymphocyte count (ALC), a blood-test metric, “hidden in plain sight,” saying immune depletion has been underappreciated despite its measurability.
Soon-Shiong also criticized major cancer drugs, citing Taxol, which is marketed by Bristol Myers, and Amgen’s Neupogen. He argued that these approaches reflected an industry-wide strategy of enabling more intensive chemotherapy and managing its side effects, rather than addressing underlying immune suppression.
Against that backdrop, he spotlighted IL-15 as the biological engine behind Anktiva, positioning it as an immune amplifier rather than a traditional therapy. He contrasted older IL-2 approaches with IL-15 biology, saying IL-15 expands natural killer cells and memory cells without boosting immunosuppressive cells.
IBRX 2025 Results Preview
ImmunityBio is set to report full-year 2025 earnings on Tuesday, with expectations for sharply higher revenue and narrowing losses as Anktiva's commercialization scales.
Fiscal AI estimates 2025 revenue of $113.29 million, up from $14.75 million in 2024, with loss per share improving to $0.38 from $0.59. The company has indicated $113 million in net product revenue for Anktiva in 2025, up 700% year over year, while fourth-quarter revenue rose 431% to $38.3 million, topping estimates. Full-year net loss attributable to common shareholders improved to $351.4 million from $413.6 million in 2024.
Regulatory Wins Bolster Anktiva Outlook
Soon-Shiong has recently amplified Anktiva’s IL-15 mechanism in national media appearances and highlighted combination approaches with checkpoint inhibitors such as Merck’s Keytruda.
ImmunityBio recently completed enrollment ahead of schedule in its Phase 2 Quilt 2.005 trial evaluating Anktiva plus BCG in early-stage bladder cancer, citing improved cancer-free rates compared with standard BCG alone. A biologics license application for the regimen is expected by the fourth quarter of 2026.
Anktiva is already approved in combination with BCG for bladder cancer in the U.S. and has received authorizations in the UK, Europe, and Saudi Arabia, where regulators also granted accelerated approval for use with a checkpoint inhibitor in metastatic non-small cell lung cancer.
How Did Stocktwits Users React?
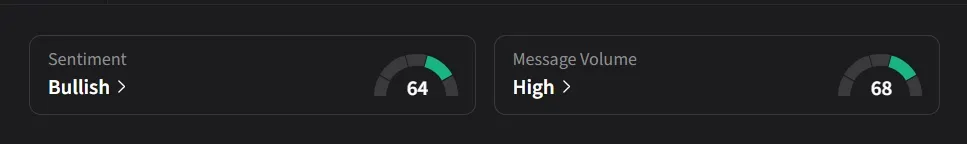
On Stocktwits, retail sentiment for IBRX was ‘bullish’ amid a 185% jump in 24-hour message volume.
One user said that, “If [broader Saudi] approval comes through, the stock could jump toward $20 quickly, and a squeeze could drive it over $100.”
Another user said, “Will be go[ing to] $11 tomorrow. Let’s bet.”
IBRX stock has surged over 400% so far this year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1214132428_1_jpg_ee44370884.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2236688965_jpg_b00d009983.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_arthur_hayes_2026_OG_jpg_9f3524f8b1.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2261265006_jpg_cd8b462617.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2256966922_jpg_81b5386f83.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_zoom_video_logo_resized_15c98df8d1.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)