Advertisement|Remove ads.
IBRX Stock Lights Up: Retail Cheers Calls For Trump To ‘Pull The Plug’ On FDA Chief Marty Makary Amid Anktiva Row

- The Wall Street Journal said patience with Makary is “wearing thin” among Republicans and White House officials, calling for Trump to “pull the plug.”
- Anktiva remains under pressure after the FDA issued a Refusal to File letter last year for its expanded use in papillary bladder cancer.
- The therapy has also faced FDA warning letters over promotional claims.
Shares of ImmunityBio, Inc. (IBRX) jumped 5% on Monday after a weekend report calling for the removal of FDA chief Marty Makary fueled speculation around the outlook for its cancer therapy, Anktiva.
IBRX stock rose marginally overnight heading into Tuesday. The stock surged 193% over the past year.
FDA Under Fire Amid Anktiva Row
The Wall Street Journal’s opinion piece by Allysia Finley over the weekend said that Makary’s tenure at the U.S. Food and Drug Administration (FDA) has been influenced by controversial drug decisions, internal clashes and political backlash. The report added: “My sources say that patience with Dr. Makary is wearing thin among Republicans in Congress and White House officials. It’s time for Mr. Trump to pull the plug on the Makary show.”
The report also highlighted inconsistent handling of cancer and rare-disease therapies, including cases where drugs were rejected or delayed despite prior regulatory talks. It also highlighted internal tensions within the FDA, including disputes involving former biologics head Vinay Prasad and turnover among senior officials overseeing drug review divisions.
Prasad, who previously led the agency’s biologics and gene therapy unit, had earlier called ImmunityBio founder Patrick Soon-Shiong a “snake oil salesman” during a 2024 episode of The VPZD Show.
Anktiva Hits Regulatory Roadblocks In US
Meanwhile, tough regulatory conditions have weighed on Anktiva, an IL-15-based immunotherapy used in combination with standard BCG therapy for certain bladder cancer patients. Last year, the FDA issued a Refusal to File (RTF) letter for a supplemental application seeking to expand its use for papillary disease, despite having encouraged the company to submit the filing.
Soon-Shiong, at the time, called for clarification, citing a “confounding inconsistency” in reviewing data from the same clinical population. The company resubmitted the filing in March with additional data, including updated efficacy results and long-term follow-up.
The therapy has also faced regulatory scrutiny over its promotional claims, with the FDA issuing warning letters for statements suggesting broader applications for cancer. Soon-Shiong pushed back publicly, saying a cited television advertisement “never aired at all” and that the issue stemmed from how scientific discussion was interpreted under promotional rules.
However, ImmunityBio’s past ties with U.S. President Donald Trump could offer upside for IBRX shares if the WSJ report unfolds. Soon-Shiong previously said discussions involving Trump and Saudi bigwigs helped push international momentum for Anktiva.
While U.S. regulatory friction has long persisted, the Saudi Food and Drug Authority has already granted accelerated approval for Anktiva, in combination with checkpoint inhibitors, for metastatic non-small cell lung cancer.
IBRX Retail Chatter Spikes On WSJ Report
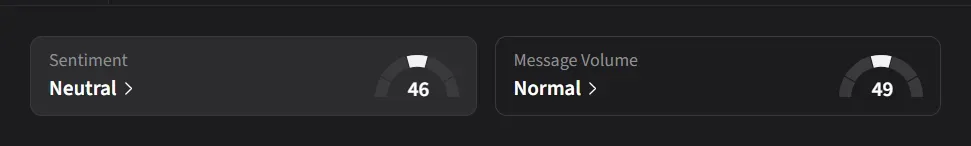
IBRX sentiment among retail traders on Stocktwits was ‘neutral’ amid a 291% jump in 24-hour message volumes as they weighed in on the WSJ report, linking the report directly to Anktiva’s regulatory outlook and broader biotech sentiment.
“But gone now. Anktiva is the test for Makary. Does Makary’s new approval paradigm win, or do Big Pharma entrenched bureaucrats win? We will know ‘Soon’!” one user said. Another user said, “Reality is, that the whole system is corrupt. Trump is ready to fire Makary (according to WSJ-who knows)…Be patient and I think there will be some government contracts sooner rather than later.”
A third user added, “I think this is the reason that many small cap bios and XBI are moving up today…Makary talks in a manner that seems bullish for these companies, but then the FDA rejects the very same drugs or fast tracks that he has promised.”
Others pointed to past regulatory friction, with one post warning, “This is also terrifying for the FDA running interference…The egg on their face if they dare try another RTF on Papillary.”
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)