Advertisement|Remove ads.
ImmunityBio Founder Pushes Back After FDA Warning Letter Over Anktiva Claims — Says Ad 'Never Aired At All'

- The FDA warning letter cited both the TV ad and Soon-Shiong’s earlier appearance on The Sean Spicer Show as suggesting uses beyond Anktiva’s approved indication.
- Soon-Shiong said the FDA classified the podcast discussion as direct-to-consumer “promotional speech” rather than scientific commentary.
- IBRX stock fell as much as 30% after the warning letter, marking the stock’s biggest intraday drop in 15 months.
ImmunityBio, Inc. (IBRX) founder Patrick Soon-Shiong said a television advertisement referenced in a recent U.S. Food and Drug Administration (FDA) warning letter over promotional claims tied to Anktiva “never aired at all,” pushing back publicly during a Monday appearance on The Sean Spicer Show.
Founder Pushes Back On Cited TV Ad
“The warning letter related to two things, one, a TV ad and two, the Sean Spicer podcast,” Soon-Shiong said, referring to promotional claims tied to Anktiva. “The TV ad, as it turned out, we never had the TV ad.” He added that regulators referenced only a transcript submitted as part of routine promotional review procedures, rather than a broadcast commercial. “The potential transcript of that ad never aired at all,” he reiterated.
Anktiva is an IL-15-based immunotherapy approved along with BCG for certain patients with non-muscle invasive bladder cancer that no longer responds to standard therapy.
The FDA warning letter, issued last month, cited both a television advertisement and Soon-Shiong’s earlier January appearance on the same Sean Spicer Show as examples of promotional language suggesting uses beyond the therapy’s approved indication for BCG-unresponsive non-muscle-invasive bladder cancer.
The regulator said statements on the advertisement and podcast “provide evidence that Anktiva is intended for new uses for which it lacks approval.” One remark flagged by the agency involved Soon-Shiong saying during the earlier interview that the therapy “actually can treat all cancers.”
The warning letter also cited language describing Anktiva as a cancer vaccine that “can treat the tumor, and it doesn’t come back,” saying such claims were misleading. Shares of IBRX fell as much as 30% following the warning letter, marking the stock’s biggest intraday decline in 15 months.
ImmunityBio’s Free Speech Tension With FDA
During Monday’s appearance, Soon-Shiong said the FDA classified the podcast discussion as direct-to-consumer promotional communication rather than scientific commentary. “The issue really was that they considered my podcast with you a promotional speech, which then fell under very strict FDA rules.”
He called the warning letter part of a broader regulatory boundary between scientific discussion and marketing for approved therapies. “There’s really a tension, a real tension between the First Amendment of free speech and the FDA’s policy of what they call promotional speech.”
He added that the health regulator mandates that discussions be strictly limited to labeled indications and safety disclosures when a drug is referenced by name in a consumer-facing setting.
Soon-Shiong said the agency indicated that the earlier interview had been brought to its attention, referencing a “Bad Ad program,” which is apparently a reporting system that allows people to flag potentially misleading prescription drug promotions to the FDA.
Founder Reaffirms Anktiva Approved Indication
Soon-Shiong used the interview to restate the therapy’s currently authorized indication and safety requirements, emphasizing that Anktiva remains approved only in combination with BCG for a specific subset of bladder-cancer patients. “Anktiva labeling includes a warning for risk of metastatic bladder cancer with a delayed cystectomy,” Soon-Shiong added.
The founder said ImmunityBio removed the earlier podcast from its website and offered to return to the program to clarify the therapy’s approved indication and safety context.
Additionally, the company submitted a formal response to regulators outlining compliance steps, including enhanced promotional-review protocols, executive training and external regulatory oversight.
How Do Retail Traders Feel About IBRX?
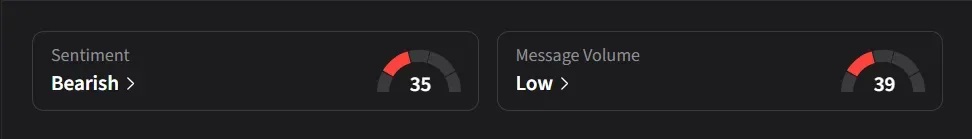
On Stocktwits, retail sentiment for IBRX has slipped to ‘bearish’ from ‘extremely bullish’ levels in the previous quarter amid a 250% surge in 24-hour message volumes.
One user said, “My personal take away from the Spicer podcast “ They are moving very very fast” Every country is moving very fast = Not slow walking like our US FDA. Moving very= growing fast. UNSTOPPABLE!! STAY TUNED!! “
Another user said, “So the FDA dropped our SP by 30% on the day with a warning letter on an add that did not even air and that was in fact sent to them in advance for approval....”
IBRX stock has jumped 178% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)