Advertisement|Remove ads.
After IOVA Stock’s Best Day In 2 Years, Analyst Sees Over 300% Upside: Here’s Why

- Chardan reiterated a ‘Buy’ rating and sees 323% upside, despite trimming its price target to $16 from $17.
- Q4 margins hit a record high, with gross margin climbing to 50% from 43% in Q3, which the interim CEO said was Iovance’s “best ever” performance.
- Pipeline momentum strengthened, with FDA Fast Track designation for Lifileucel in nonsquamous lung cancer.
Advertisement|Remove ads.
Shares of Iovance Biotherapeutics, Inc. (IOVA) posted their best session in two years on Tuesday after the company reported record fourth-quarter (Q4) margins, with investor sentiment buoyed by an analyst rating that implied more than 300% upside from current levels.
IOVA stock jumped over 30% to $3.78 in Tuesday’s regular session, and added another 0.7% in extended trading.
Chardan Sees Huge Upside For IOVA
On Tuesday, Chardan lowered its price target on Iovance to $16 from $17, implying a 323% upside from current levels, while retaining a ‘Buy’ rating.
Advertisement|Remove ads.
The brokerage attributed the Q4 margin rise to operational optimization and said further improvements are expected through 2026 as the company internalizes manufacturing of Lifileucel, its immune cell therapy for solid tumors, at its iCTC facility and optimizes research and development spending. It added that Iovance’s pipeline continues to advance, with early data in soft tissue sarcomas “looking encouraging.”
Amtagvi Drives Strong Q4 Revenue Surge
Iovance reported Q4 revenue of $86.7 million, beating analyst expectations of $81 million, while posting a Q4 loss of $0.18 per share, in line with estimates.
Q4 total product revenue rose about 30% from the previous quarter to $87 million, led by about $65 million in U.S. Amtagvi revenue, a cell-based therapy for advanced melanoma, and $22 million from global Proleukin sales, a drug that helps activate the immune system during treatment.
Advertisement|Remove ads.
Gross margin from cost of sales climbed to 50%, up from 43% in the prior quarter, marking the company’s “best ever” margin performance, according to interim CEO Frederick Vogt, as said on the earnings call.
For full-year 2025, Iovance reported total product revenue of about $264 million, coming in within its guidance range of $250 million to $300 million. The company ended the year with $303 million in cash, which is expected to fund operations into the third quarter of 2027.
CEO Sees “Remarkable” Revenue Growth In 2026
Vogt said Iovance achieved its 2025 guidance range in its first full year of commercial launch, driven by rising demand for Amtagvi. On the earnings call, he said the company expects “remarkable revenue growth” in 2026 and sees potential for Amtagvi and Proleukin to generate more than $1 billion in peak U.S. sales.
Advertisement|Remove ads.
The company noted Amtagvi’s approval in Canada and pending regulatory decisions in the U.K. and Australia in the first half of 2026, with Switzerland targeted for the first half of 2027. Iovance is also working with European regulators to resubmit a marketing authorization application in 2026.
FDA Fast Track Granted For Lifileucel In Lung Cancer
Iovance said Lifileucel has received Fast Track Designation from the FDA for previously treated nonsquamous non-small cell lung cancer, with interim data showing a 26% objective response rate and durability extending beyond 25 months. The company expects to complete enrollment in 2026 and to target an accelerated launch in the second half of 2027.
Separately, the company announced positive early data in advanced soft-tissue sarcomas, where Lifileucel delivered a 50% confirmed response rate in an initial cohort of heavily pretreated patients. Iovance plans to initiate a single-arm registrational trial in the second quarter of 2026 and engage with regulators on an accelerated approval pathway.
Advertisement|Remove ads.
How Did Stocktwits Users React?
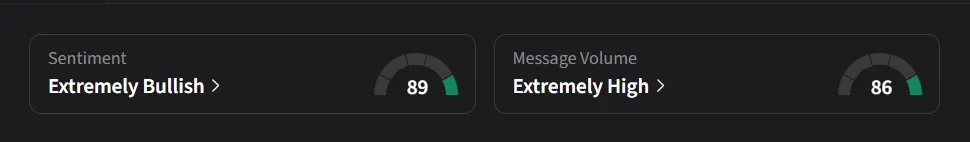
On Stocktwits, retail sentiment for IOVA was ‘extremely bullish’ amid a 550% surge in 24-hour message volume.
One user said, “potentially this could help Q1 margins go up further. If they can reach somewhere around 60%, it’ll be great progress towards profitability.”
Advertisement|Remove ads.
Another user said, “THIS IS A 10/ share stock no doubt. You still have time to get it and make hay.”
IOVA stock has surged 38% year to date.
Advertisement|Remove ads.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_strategy_logo_OG_jpg_fa4e1a7d04.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1376765653_jpg_d56a66290e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275170769_jpg_e811c3ae51.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spacex_jpg_6eb3abcb72.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2271060260_jpg_b0369496dd.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Marvell_jpg_2d5ebc76bc.webp)