Advertisement|Remove ads.
MLTX Stock Soars Overnight After FDA Backs Skin Drug Filing — Analyst Sees Over 300% Upside On ‘Best-In-Class’ Label

- MoonLake said that the FDA agreed to include results from both the Phase 2 Mira study and Phase 3 Vela trials in the proposed drug label.
- Clear Street maintained a ‘Buy’ rating and $70 price target on MLTX, implying a 310% upside from current levels.
- MoonLake ended the first quarter with $357.9 million in cash and said its current funding runway extends through the end of 2027.
Shares of MoonLake Immunotherapeutics, Inc. (MLTX) jumped 7% in overnight trading heading into Monday after the company said that the U.S. Food and Drug Administration (FDA) aligned on its approval filing plans for Sonelokimab, prompting Clear Street to say that the skin therapy could emerge with a “stronger, best-in-class label.”
MLTX stock snapped two straight weeks of weekly losses and ended 2% higher last week at $17.06.
FDA Backs MoonLake HS Drug Filing
On Sunday, MoonLake said that its final pre-biologics license application (BLA) meeting with the FDA concluded with alignment on certain parts of its application for Sonelokimab in hidradenitis suppurativa (HS), a chronic inflammatory skin disease.
MoonLake said that the FDA agreed the results from both its mid-stage Mira study and late-stage Vela trials can be included in the proposed prescribing label for Sonelokimab, boosting the drug’s case for approval in HS. The company also said that the FDA supported including data from the Vela-Teen study, which could allow the treatment to be approved for patients aged 12 and older.
The company plans to apply for a Priority Review designation and expects to submit the BLA application by September. Acceptance of the filing, including a decision on Priority Review status, is expected by November. MoonLake also said that the FDA identified no remaining gaps, including manufacturing-related issues, that would prevent the filing.
MoonLake said that the proposed label could highlight that 43% of patients in the Mira study achieved a high level of symptom improvement after 12 weeks of treatment, with results outperforming placebo by 29 percentage points. According to the company, the efficacy levels are the strongest reported so far in any well-controlled late-stage clinical study for HS.
Clear Street Sees ‘Best-In-Class’ Label
After the FDA meeting, Clear Street said that MoonLake’s positive pre-application meeting restores the company to its “prior strong positioning” in the HS market. The brokerage said that the FDA’s agreement to include both Mira efficacy data and Vela trial results in the label could result in a “stronger, best-in-class label” for Sonelokimab.
Clear Street maintained a ‘Buy’ rating on the stock with a $70 price target, implying an upside of 310% from current levels.
MLTX Cash Runway Extends Into 2027
As of March 31, MoonLake held $357.9 million in cash, cash equivalents and short-term marketable securities, which it expects to fund operations and spending through the end of 2027.
The company also retains access to up to $400 million in additional non-dilutive funding through its Hercules Capital debt facility. Research and development expenses totaled $54.5 million during the first quarter, compared with $56 million in the previous quarter. General and administrative expenses rose to $15.5 million from $9.2 million in the prior quarter. MoonLake said the increase was mainly from $4.8 million in accelerated expense recognition related to the voluntary cancellation of unvested stock option awards.
MLTX Reports Strong Long-Term HS Results
The FDA update comes on the heels of encouraging long-term clinical data reported by MoonLake from the Phase 3 Vela-1 and Vela-2 trials in HS. In March, the company said 62% of patients treated with Sonelokimab achieved strong clinical improvement after 40 weeks, while up to 32% saw their skin lesions become nearly clear.
Up to 25% of patients achieved inflammatory remission, meaning that the key signs of the disease disappeared completely. Patients also reported improvements in pain, mobility and daily activities such as walking and dressing. According to the company, up to 43% of patients saw reductions in their worst skin pain, while 65% reported improvements in overall quality of life.
More Trial Catalysts Ahead For MLTX
Earlier this year, MoonLake reported positive mid-stage trial results for Sonelokimab in axial spondyloarthritis, an inflammatory condition that affects the spine and joints. The company said that 81% of patients showed improvement within 12 weeks of treatment, with scans and disease markers also indicating reduced inflammation and disease activity.
Investors are now watching several upcoming catalysts over the next year, including 52-week data from the Phase 3 Vela studies in the second quarter, results from the Phase 3 Izar-1 trial in psoriatic arthritis and data from the Vela-Teen adolescent study in mid-2026, followed by the BLA submission for HS by the end of September.
How Do Retail Traders Feel About MLTX?
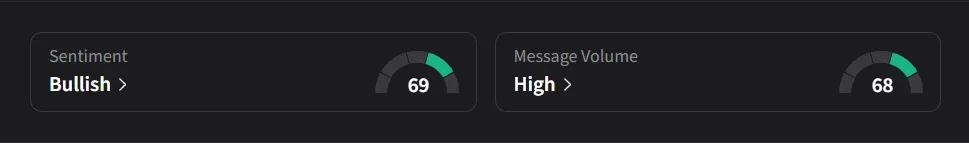
On Stocktwits, retail sentiment for MLTX flipped to ‘bullish’ from ‘bearish’ levels a day ago amid nearly a 400% surge in 24-hour message volumes.
One user said, “Looking forward to tomorrow! We’ve already been through a lot with this stock. But with this news, the selling pressure should be very low, and it should be possible to break through the USD 20 . After that, it’ll be easier to move higher.”
Another user said, “There's literally nothing standing in SLK's path to becoming Best in Class across all Inflammatory skin disease indications. Furthermore, we have zero concern of dilution which almost always happens during phase 3 trials nearing drug BLA submission.”
MLTX stock has declined 57% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: Bitcoin Flips Tesla Again In Trillion-Dollar Tug-Of-War

/filters:format(webp)https://news.stocktwits-cdn.com/large_Big_Bear_jpg_8fce0f24aa.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2252565019_jpg_dba152319e.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263898051_jpg_9e75888009.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2197271762_jpg_e7153010f5.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2249184989_jpg_2f36d82a30.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_950698324_jpg_d6201b2562.webp)