Advertisement|Remove ads.
MLTX Stock Jumps Overnight: Lead Skin Disease Drug Delivers Strong Phase 3 Gains With Majority Of Patients Improving

- The company's week-40 Phase 3 data showed continued improvement in most patients treated with Sonelokimab for hidradenitis suppurativa.
- About 62% of patients showed strong improvement after 40 weeks of treatment, with up to 32% seeing near-complete lesion clearance.
- Up to 25% hit inflammatory remission, while patients also reported reductions in pain and improvements in daily functioning.
Shares of MoonLake Immunotherapeutics, Inc. (MLTX) jumped over 6% in overnight trading late Sunday after updated Week-40 Phase 3 data showed continued improvement in most patients treated with Sonelokimab, its lead therapy for hidradenitis suppurativa, an inflammatory skin disease.
MLTX stock slipped over 2% on Friday but is still on track to post its second straight quarterly gain.
Sonelokimab Shows Strong Week-40 Gains
New data from the Vela-1 and Vela-2 trials showed that 62% of patients treated with Sonelokimab had strong improvement after 40 weeks, while up to 32% saw their skin lesions nearly clear. Up to 25% achieved inflammatory remission, meaning the key signs of the disease disappeared completely.
Patients also reported meaningful improvements in pain, movement and everyday activities such as walking and getting dressed. Up to 43% experienced a clear reduction in their worst skin pain, and 65% reported overall improvements in quality of life. The treatment’s safety profile remained consistent with earlier studies, with no new safety concerns reported.
“The Week 40 results from the VELA trials show an early and increasing clinical benefit over time for patients treated with Sonelokimab, addressing a critical goal in HS treatment: long-term disease control,” said Alexa Kimball, CEO of Harvard Medical Faculty Physicians at Beth Israel Deaconess Medical Center.
Momentum Builds Across Indications
The update adds to growing momentum for Sonelokimab following positive Phase 2 results last month in axial spondyloarthritis, an inflammatory spine disease.
In the S-Olaris trial, 81% of patients showed improvement within 12 weeks, supported by imaging evidence of reduced joint inflammation and biomarker signals linked to disease activity. The safety profile remained consistent with prior studies.
Runway Extends Ahead Of 2026 Catalysts
MoonLake ended 2025 with $394 million in cash and marketable securities, alongside proceeds from a recent $75 million equity raise, which the company expects will support operations into the second half of 2027. It also expanded its Hercules Capital facility, providing access to up to $400 million in additional non-dilutive funding.
Investors are now watching several catalysts this year, including 52-week data from the Phase 3 Vela trials in hidradenitis suppurativa in the second quarter, results from the Phase 3 Izar-1 trial in psoriatic arthritis in mid-2026, a biologics license application submission for hidradenitis suppurativa in the second half of 2026, and results from the Phase 3 Izar-2 trial later in the second half of 2026.
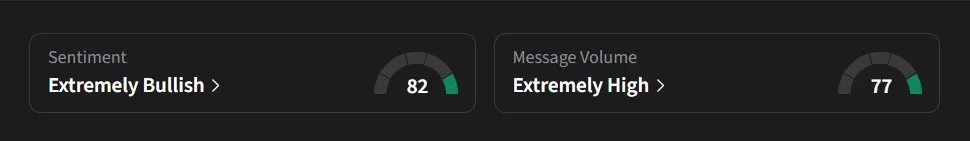
What Is The Retail Mood On Stocktwits?
On Stocktwits, retail sentiment for MLTX was ‘extremely bullish’ amid nearly an 800% surge in message volumes over the past week.
One user said, “MLTX's SLK is now the "Gold Standard" in HS treatment if a doctor actually cares about curing this debilitating disease!”
Another user pointed to the trial results and said, “This is going to be GREAT!!! But Monday is going to be volatile with all the shorting hedgehogs and gamma chasers trying to reset their positions.”
MLTX stock has declined nearly 60% over the past year, but is up over 30% since January.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: March’s Almost Over — Can Bitcoin Still Meet Tom Lee’s April Call?

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2253111144_jpg_12be2fed14.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_6979_jpg_a2a1032fdc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Jensen_Huang_jpg_8579a8a271.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/AMD_s_Lisa_Su_1200pi_gettyimages_resized_jpg_2b6947251d.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2274484367_jpg_e7062348b5.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263602546_jpg_a0f4e7e3ca.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2227884315_jpg_baa8a96bba.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)