Advertisement|Remove ads.
SLS Stock Slips After-Hours As Cancer Pipeline Silence Frustrates Retail — China Licensing Fight Now In Spotlight

- Anticipated updates on the company’s leukemia drug SLS009 did not land, dampening near-term catalyst momentum.
- Earlier studies showed SLS009 triggered leukemia cell death and remained active in difficult-to-treat AML cases with ASXL1 and TP53 mutations.
- Attention also remains on the Phase 3 Regal trial, where survival events are approaching the threshold required to begin topline analysis.
Advertisement|Remove ads.
Shares of Sellas Life Sciences Group, Inc. (SLS) slipped nearly 1% in after-hours trading on Tuesday as investors remained in wait-and-watch mode after expected cancer-pipeline updates failed to land, turning focus toward a pending China licensing arbitration decision tied to Phase 3 milestone payments for its lead acute myeloid leukemia (AML) program.
SLS stock ended the regular session about 1% higher at $5.03.
SLS009 Update Fails To Land
Investor expectations had been building around potential updates on the company’s leukemia drug SLS009, which earlier studies showed could help trigger cancer cell death while blocking survival signals that leukemia cells rely on. The treatment also showed activity in hard-to-treat AML cases with ASXL1 and TP53 mutations, genetic changes often linked to poorer outcomes.
Advertisement|Remove ads.
SLS shares also drew investor attention on Tuesday after U.S. investment manager Dagco increased its stake in the company by 78% ahead of anticipated catalysts in the company's leukemia pipeline.
China Milestone Payment Dispute In Focus
Focus has now turned to an arbitration proceeding involving Sellas and its Chinese partner 3D Medicines covering development and commercialization rights for 3D189, the regional version of the company’s immunotherapy candidate Galinpepimut-S (GPS), as well as 3D059, the regional version of SLS009.
The arbitration includes disputes over milestone payments tied to the initiation of Phase 3 clinical development for 3D189, and whether the regional partner maintained sufficient expertise and resources to meet its obligations under the licensing agreement.
Advertisement|Remove ads.
The tribunal directed both parties to submit final closing submissions by April 21, moving the case into its decision phase.
Sellas previously said that it received $10.5 million in upfront and milestone payments under its Greater China agreement with 3D Medicines, while up to $191.5 million in additional development, regulatory and commercial milestone payments remain possible under the licensing deal, excluding potential future royalties.
Regal Trial Readout Watch Builds
Beyond the arbitration process, investor attention continues to center on the company’s ongoing Phase 3 Regal trial evaluating GPS as a maintenance therapy for AML patients whose disease returned but later entered remission after additional therapy.
Advertisement|Remove ads.
Sellas previously said 72 overall survival events had been recorded as of late December, approaching the 80-event threshold required to begin topline analysis. CEO Angelos Stergiou has said that slower accumulation of survival events in oncology trials can sometimes indicate patients are living longer than expected compared with historical assumptions.
However, with no meaningful update emerging from the summit session as expected, attention appears to shift toward another near-term catalyst for the drugmaker.
How Do Retail Traders Feel About SLS?
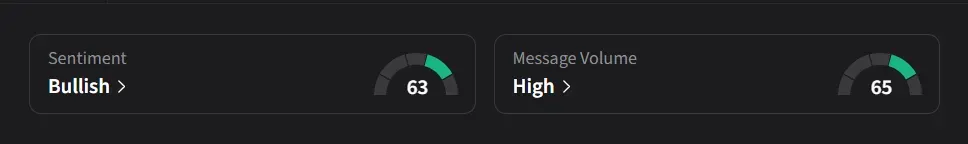
On Stocktwits, retail sentiment for SLS has improved to ‘bullish’ from ‘neutral’ levels a day ago amid ‘high’ message volumes.
Advertisement|Remove ads.
One user noted that while the SLS009 poster presentation drew attention, the absence of a formal company update or management communication on National AML Awareness Day stood out.
Another user expressed frustration, saying, “If they are going to go silent then do that and don't go to conferences, don't tweet, stop communicating completely. It just gives Shorts something else to see. Sterg we don't want to hear from you unless it is a BO.”
Advertisement|Remove ads.
SLS stock has surged 290% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Advertisement|Remove ads.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_cannabis_generic_b5230b29c3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_Trade_desk_logo_resized_c0229eb2ab.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2255802445_jpg_427b92fbb5.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_fuelcellenergy_resized_ef1a57cfe6.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_oscar_health_jpg_89aa74c6fe.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2217651425_jpg_0d7dc8a6cd.webp)