Advertisement|Remove ads.
LTRN Stock Gains 18% Pre-Market: FDA Greenlights First Pediatric Cancer Trial For Oncology Compound

- STAR-001 is a precision oncology compound discovered using Lantern’s proprietary RADR AI platform.
- The study will evaluate the drug both as a standalone treatment and in combination with Spironolactone.
- The trial to be conducted with the Pediatric Oncology Experimental Therapeutics Investigators' Consortium.
Shares of Lantern Pharma Inc. (LTRN) surged nearly 18% in pre-market trading on Friday after the biotech firm said that the U.S. Food and Drug Administration cleared its Investigational New Drug (IND) application for STAR-001, allowing the company to begin a Phase 1 pediatric clinical trial for central nervous system cancers.
STAR-001 is a precision oncology compound discovered using Lantern’s proprietary RADR AI platform, which helped identify its potential in CNS and pediatric CNS indications, the company said. The study will evaluate the drug both as a standalone treatment and in combination with Spironolactone in children with relapsed or refractory CNS cancers.
The trial will be conducted in collaboration with the Pediatric Oncology Experimental Therapeutics Investigators' Consortium.
Lantern's Drug To Treat Soft Tissue Sarcomas
Earlier this year, the FDA granted Orphan Drug Designation to Lantern Pharma's experimental drug LP-284 for the treatment of soft tissue sarcomas. It marked the third orphan designation for LP-284 and the sixth across the company’s pipeline.
Orphan designation provides incentives to support the development of treatments for rare diseases affecting fewer than 200,000 people in the U.S.
LP-284 is a small-molecule therapy designed to target cancer cells with DNA repair defects. The drug has shown encouraging activity in early studies, including a complete response in a heavily pretreated lymphoma patient in a Phase 1 trial.
The therapy is currently being tested in patients with B-cell non-Hodgkin lymphomas.
How Did Stocktwits Users React?
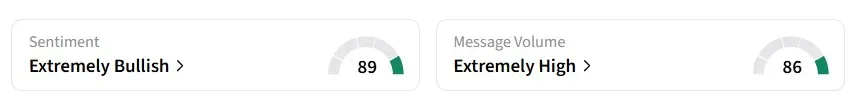
Retail sentiment for LTRN on Stocktwits turned 'extremely bullish' from 'bullish' a day earlier, amid 'extremely high' message volumes.
The stock has been under heavy selling pressure, slumping more than 50% in 2026.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Trending_stock_resized_may_jpg_bc23339ae7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275396018_jpg_a205b2aecb.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275353529_jpg_271d3f9c9b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2238553113_jpg_02cf1c9b5e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2235501291_jpg_6911a5be03.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_6979_jpg_a2a1032fdc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Stock_chart_representative_image_resized_jpg_dacf5b1590.webp)