Advertisement|Remove ads.
CAPR Stock Gains Steam: Former Capricor Exec Could Become FDA’s Top Gene Therapy Regulator Ahead Of High-Stakes DMD Decision

- Hemmati served as Capricor’s vice president of medical and clinical development for new therapies between 2015 and 2017.
- Capricor is awaiting an FDA decision on its DMD therapy, Deramiocel, expected by Aug. 22.
- The FDA resumed review last month after issuing a Complete Response Letter in July 2025, requesting additional clinical data.
Shares of Capricor Therapeutics (CAPR) jumped 1% in premarket trading on Tuesday after reports said a former company executive is being considered to lead the FDA’s top gene therapy division just months before a key approval decision on its Duchenne muscular dystrophy (DMD) treatment.
CAPR stock jumped 11% on Monday to end at $34.54, logging its best session in nearly two months.
Former Capricor Executive Emerges As FDA Biologics Chief Candidate
Houman Hemmati, who served as vice president of medical and clinical development for new therapies at Capricor between 2015 and 2017, is among the candidates being interviewed by FDA Commissioner Marty Makary to head the Center for Biologics Evaluation and Research, or CBER, Bloomberg reported.
The division oversees approvals of vaccines and advanced gene and cell therapies, including treatments for rare diseases such as DMD. However, HHS spokesperson Andrew Nixon reportedly said no selection has been finalized and that the agency is still evaluating candidates.
CAPR Awaits Key FDA Ruling On DMD Therapy
The development comes as Capricor awaits an FDA decision on Deramiocel, its investigational therapy for DMD, a rare genetic condition that causes progressive muscle weakness in children, expected by Aug. 22.
The FDA resumed review of Deramiocel last month after issuing a Complete Response Letter (CRL) in July 2025, which indicated earlier that the agency could not approve the drug in its current form and typically requests additional data. Capricor’s resubmission included Phase 3 Hope-3 trial results showing 54% slower deterioration in upper-limb function and 91% slowing of cardiac disease progression, both statistically significant compared with placebo.
Deramiocel consists of donor-derived cardiac cells, which reduces inflammation linked to muscle degeneration in DMD.
Prasad Exit Seen As Positive For Biotech Approvals
The CBER leadership opening follows the departure this month of outgoing director Vinay Prasad, whose tenure drew criticism from the biotech industry over regulatory decisions affecting rare-disease therapies.
Prasad’s exit news lifted several biotech stocks last month, with BMO Capital Markets calling the change “a sigh of relief” for the sector and saying it could improve approval prospects for multiple companies developing advanced therapies.
During his tenure, Prasad was involved in disputes with companies, including Sarepta Therapeutics, over gene-therapy reviews and disagreements with UniQure and Regenxbio. He also briefly stepped away from the agency last year amid political criticism over regulatory decisions before returning weeks later with Makary’s support.
How Do Retail Traders Feel About CAPR?
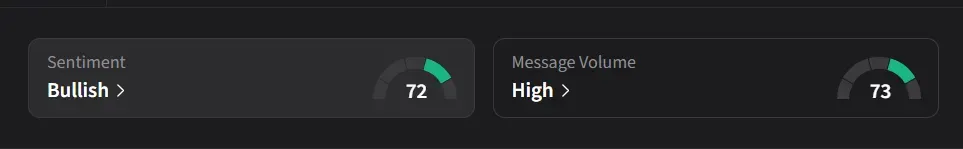
On Stocktwits, retail sentiment for CAPR jumped to ‘bullish’ from ‘bearish’ levels over the past day amid ‘high’ message volume.
One user said, “$45-50 is my guess if the appointment is confirmed. 75+ upon approval, and $100+ with sale numbers,” referring to the prospects of Hemmati leading the CBER.
Another user said, “if Dr Hemmati gets appointed as CBER chief, Vinay Prasad is gonna want credit for CAPRs approval. Will beat David to the punch. Guaranteed! Lol”
CAPR stock has jumped over 270% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_492469887_jpg_1b932ad0fb.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Satellite_internet_communication_jpg_11d7fa9a96.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_marvell_logo_OG_jpg_dfc748dd9b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Chinmay_Rautmare_Photo_jpeg_96dd37efd0.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275088577_jpg_0ce4079eb7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_6979_jpg_a2a1032fdc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Chamath_Palihapitiya_OG_jpg_c607fb4198.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Trending_stock_resized_may_jpg_bc23339ae7.webp)