Advertisement|Remove ads.
Capricor Therapeutics’ DMD Treatment Study Shows Improvement In Key Heart Function Measures

- MRI results highlighted that patients with baseline cardiomyopathy saw a 3.3 percentage-point improvement in left ventricular ejection fraction.
- Capricor has already submitted a BLA for Deramiocel to the FDA.
- The company said the findings were presented at the 2026 Muscular Dystrophy Association Clinical & Scientific Conference in Orlando.
Capricor Therapeutics (CAPR) was in focus on Thursday after the biotech firm said new data from its Phase 3 HOPE-3 trial showed that its therapy, Deramiocel, improved key heart function measures in patients with Duchenne muscular dystrophy (DMD).
The findings were presented at the 2026 Muscular Dystrophy Association Clinical & Scientific Conference in Orlando.
CAPR stock fell around 1.5% on Thursday.
Endpoint Indicates Overall Treatment Benefit
Cardiac MRI showed a significant reduction in fibrosis in patients treated with Deramiocel after 12 months compared with placebo. The MRI results also highlighted that patients with baseline cardiomyopathy saw a 3.3 percentage-point improvement in left ventricular ejection fraction.
Researchers reported a statistically significant overall treatment benefit using a composite measure of patient function and heart health.
FDA Decision Expected In August
Capricor has already submitted a Biologics License Application (BLA) for Deramiocel to the U.S. Food and Drug Administration (US FDA).
The application is currently under review, and the regulator has set Aug. 22, 2026, as the decision date.
Earlier this week, the FDA lifted a previous Complete Response Letter and resumed review of its BLA, moving the therapy closer to potential approval. Capricor expects to be eligible for a Priority Review Voucher (PRV) if Deramiocel receives FDA approval.
How Did Stocktwits Users React?
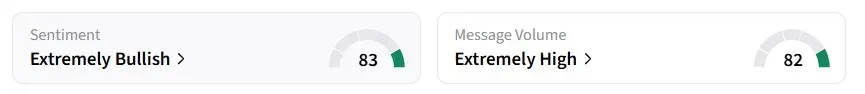
Retail sentiment on Stocktwits remained in the ‘extremely bullish’ territory over the past 24 hours, accompanied by ‘extremely high’ message volumes.
One user advised ignoring any noise that could interfere with reasonable expectations.
Another user said the results showed the drug is actually repairing the heart, not just slowing decline.
The stock has gained around 14% so far this year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_kevin_o_leary_OG_jpg_2789641a97.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Bitcoin_eb6767ae0b.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2259655319_jpg_e7f29c8883.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2252501501_jpg_fe05bb3647.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_nio_stock_jpg_770e12377f.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)