Advertisement|Remove ads.
Cingulate Stock Jumps Overnight: US Patent Office Clears ADHD Drug Filing Ahead Of May FDA Catalyst

- The USPTO cleared a patent filed in November 2023, with a status indicating it has been approved and the patent is expected to be issued.
- The filing covers drug-delivery technologies for neurological treatments, including ADHD.
- The technology supports Cingulate’s Precision Timed Release (PTR) platform, used in CTx-1301, an ADHD drug.
Advertisement|Remove ads.
Shares of Cingulate, Inc. (CING) jumped over 4% in overnight trading on Sunday after the U.S. Patent and Trademark Office (PTO) cleared a key patent application tied to the company’s ADHD drug platform, adding another potential catalyst ahead of a major regulatory decision expected in May.
CING stock snapped four straight sessions of gains on Friday to end nearly 5% lower at $8.69.
Patent Office Clears Cingulate ADHD Drug Patent
Patent records show the USPTO has allowed a patent titled “Trimodal, Precision-Timed Pulsatile Release Tablet.” The application, filed in November 2023, is now listed with the status “Allowed - Notice of Allowance Not Yet Mailed” as of Sunday. An allowed status means the patent examiner has approved the claims, and the patent will be issued once final administrative steps are completed.
Advertisement|Remove ads.
The inventors listed on the filing include Matthew Brams, Cingulate’s chief medical officer, Raul Silva, its chief science officer, and Arthur Straughn, a co-founder of the company. It also names Cingulate as the applicant. The application covers drug-delivery technologies classified for neurological treatments, including attention-deficit/hyperactivity disorder (ADHD).
How Cingulate’s ADHD Drug Technology Works
The patent application centers on a trimodal pulsatile-release tablet that releases medication at multiple timed intervals throughout the day. The tech aligns with Cingulate’s Precision Timed Release (PTR) platform, which the company is using to develop next-generation treatments for ADHD and other neurological conditions.
The approach aims to address common challenges with stimulant medications, including midday rebound and the need for multiple doses. CTx-1301 uses dexmethylphenidate, a stimulant already approved to treat ADHD, delivered through Cingulate’s Precision Timed Release platform. It releases dexmethylphenidate in three timed pulses, with the final dose delivered around the time when many extended-release stimulant medications begin to wear off. The company said the design aims to provide rapid symptom relief in the morning and consistent symptom control into the evening.
Advertisement|Remove ads.
In 2024, the company received a European patent for its CTx-1301, which covers up to 30 European territories, including the UK. The company also holds patents in Australia, Canada, and Israel.
FDA Decision Looms For ADHD Drug
The patent development comes ahead of a key regulatory milestone for Cingulate’s lead program. In October 2025, the U.S. Food and Drug Administration (FDA) accepted the company’s New Drug Application for CTx-1301 for the treatment of ADHD in children and adults. The agency set a Prescription Drug User Fee Act (PDUFA) target action date of May 31.
The application is being reviewed under a regulatory pathway that allows companies to reference existing data for approved ingredients while showing new clinical benefits through different delivery technologies. Cingulate completed Phase 3 trials in adult and pediatric patients before submitting the application. The studies showed improvements in ADHD symptoms and demonstrated sustained efficacy across the day.
Advertisement|Remove ads.
How Did Stocktwits Users React?
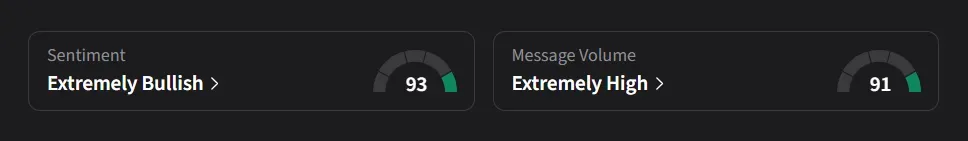
On Stocktwits, retail sentiment for CING shifted to ‘extremely bullish’ from the ‘bullish’ zone over the past month amid a 675% surge in 24-hour message volume.
One user said, “Can’t wait for Monday, interesting week ahead of us.”
Advertisement|Remove ads.
Another user said, “Fuel up the rockets, time to ride to moon.”
CING stock has doubled so far this year.
Advertisement|Remove ads.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_stock_chart_rising_resized_6ebc3dd7e4.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1500703659_jpg_805d718d3e.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_traders_jpg_94ea56f3bb.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2252501501_jpg_fe05bb3647.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2267825783_jpg_1b90bf709b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)