Advertisement|Remove ads.
Does TARA Stock’s 15% After-Hours Slump Signal A Bargain After New Bladder Cancer Data?

- The company reported strong efficacy and a clean safety profile from its ongoing Phase 2 Advanced-2 bladder cancer trial.
- Durability signals were notable, with 68% of BCG-unresponsive patients and 66% of BCG-naive patients cancer-free at six months.
- Safety and tolerability remained favorable, with mostly mild, short-lived side effects and no severe treatment-related adverse events.
Advertisement|Remove ads.
Shares of Protara Therapeutics, Inc. (TARA) fell nearly 15% in extended trading on Monday, even as the company reported strong response rates and favorable tolerability in its ongoing Phase 2 bladder cancer trial.
TARA stock jumped 9% on Monday’s regular session to $7.43, hitting its highest levels in three months and its best session in nearly a month.
Phase 2 Trial Shows Strong Efficacy Signals
The after-hours move followed updated interim results from Protara’s open-label Phase 2 Advanced-2 trial, which is testing TARA-002 as a bladder-delivered treatment in patients with high-risk non-muscle invasive bladder cancer. The study includes patients whose cancer has returned despite standard BCG therapy, as well as patients who have not previously received BCG, a commonly used bladder cancer treatment.
Advertisement|Remove ads.
Among patients whose cancer recurred after standard BCG treatment, 66% of the 35 patients evaluated achieved a complete response at some point during the study. Among those assessed at six months, 68% were cancer-free, while 33% remained cancer-free at 12 months. Statistical estimates showed a 71% probability of maintaining a complete response for at least six months, and 62% of patients who required re-treatment later achieved a complete response within six months.
In patients who had not previously received BCG, 72% achieved a complete response at some point, with 66% cancer-free at six months and 58% at 12 months. Statistical estimates indicated a 73% probability of maintaining a response for at least six months, and 67% of re-treated patients achieved a complete response by six months.
Safety Profile Remains Clean
The company also said the trial noted a favorable safety and tolerability profile for TARA-002. Most treatment-related adverse events were Grade 1 and transient, with no Grade 3 or higher events reported and no treatment-related serious adverse events observed. No patients discontinued treatment due to adverse events.
Advertisement|Remove ads.
“In addition to demonstrating impressive efficacy and safety, TARA-002 overcomes the limitations of existing NMIBC treatments that burden patients as well as urologists and their practices,” said CEO Jesse Shefferman.
The most commonly reported side effects included dysuria, bladder spasm, fatigue and urinary urgency, with bladder irritation typically resolving within hours to a few days after administration.
Analysts Remain Bullish Despite Selloff
Despite the after-hours decline, analyst sentiment toward the stock remains positive. Koyfin estimates show a 12-month average price target of $25.5, implying a potential upside of more than 240% from current levels.
Advertisement|Remove ads.
The stock carries a consensus rating of ‘Strong Buy’, with seven covering analysts. Of them, one rates the stock a ‘Strong Buy’, while six rate it ‘Buy.’
Advanced-3 Trial Planned for 2026
Protara said it expects to complete enrollment in the Advanced-2 trial cohort of patients whose cancer returned after standard BCG treatment in the second half of 2026. Enrollment in the cohort of patients who had not previously received BCG is already complete, with 31 patients enrolled.
The company also said it remains on track to initiate the Advanced-3 registrational trial in patients who have not previously received BCG in the second half of 2026, positioning TARA-002 for potential late-stage development across multiple high-risk non-muscle invasive bladder cancer populations.
Advertisement|Remove ads.
How Did Stocktwits Users React?
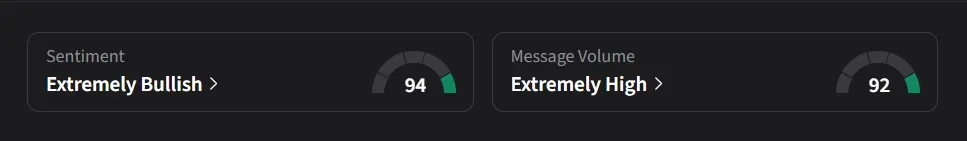
On Stocktwits, retail sentiment for TARA was ‘extremely bullish’ amid ‘extremely high’ message volume.
One user said, “I am not sure why this stock did a nose dive after hours unless the results were widely misinterpreted.”
Advertisement|Remove ads.
Another user said, “Don't catch this falling knife!! buy into tara low,” and added that investors can expect a profit on Tuesday.
TARA stock has risen 40% year to date.
Advertisement|Remove ads.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2219348572_jpg_4aa617b1f0.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/gettyimages_2251508587_594x594_1_jpg_786b90b502.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Ram_83262cba1d.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_US_economy_representative_image_jpg_88c3aa4736.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2273565031_jpg_034d495c28.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2233517117_jpg_13b62b29bf.webp)