Advertisement|Remove ads.
HELP Stock Tanks 22% Despite Strong Phase 2 Results For Anxiety Treatment: Retail Buys The Dip Ignoring Market’s Pessimism

- The clinical trial included 36 participants who continued taking existing medications for generalized anxiety disorder while receiving HLP004 injections.
- Researchers administered two doses three weeks apart and monitored participants for 12 weeks.
- Six months after treatment, about 67% of the combined study group met the criteria for a clinical response.
Helus Pharma (HELP) announced encouraging early-stage clinical results for its experimental anxiety treatment HLP004 on Thursday, reporting notable symptom improvement among patients with moderate-to-severe generalized anxiety disorder.
The clinical-stage drug developer said its Phase 2 signal detection trial showed statistically meaningful improvements when the therapy was used alongside standard antidepressant treatments.
Trial Demonstrates Significant Anxiety Reduction
In the study, participants receiving a 20-milligram dose of HLP004 alongside standard-of-care treatment experienced an average drop of roughly 10.4 points on the Hamilton Anxiety Rating Scale after six weeks.
The clinical trial included 36 participants who continued taking existing medications for generalized anxiety disorder while receiving HLP004 injections. Researchers administered two doses, three weeks apart, and monitored participants for 12 weeks, with additional follow-up planned for up to 1 year.
Helus Pharma stock traded over 22% lower in Thursday’s remarket.
Long-Lasting Response Observed In Many Patients
The company also reported sustained benefits beyond the initial treatment period. Six months after treatment, about 67% of the combined study group met the criteria for a clinical response, while 39% reached remission.
Among those receiving the higher 20-milligram dose, 59% showed improvement and 32% achieved remission after six weeks. In the lower-dose group, about 30% of patients met response and remission benchmarks at the same time point.
What Are Stocktwits Users Saying?
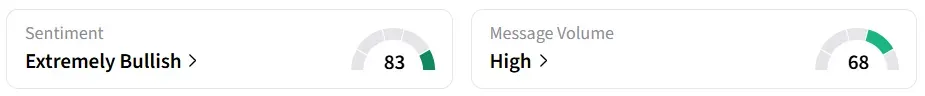
Despite the stock’s decline in the premarket session, retail sentiment on Stocktwits around the stock flipped to ‘extremely bullish’ from ‘neutral’ territory the previous day. Message volume shifted to ‘high’ from ‘normal’ levels in 24 hours.
A Stocktwits user said they have bought the dip.
HELP stock has gained over 4% year-to-date.
Also See: Liquidia Stock Gains Premarket On Q4 Revenue Beat – Investors Are Betting On YUTREPIA Growth
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)