Advertisement|Remove ads.
IBRX Stock Enters Heavy Catalyst Phase: Traders Brace For Key Anktiva Decision Amid Trump’s FDA Turmoil

- The FDA’s standard 60-day review window for ImmunityBio’s resubmitted application seeking expanded approval for Anktiva expires Thursday.
- Wall Street expects a 15% sequential jump in Q1 revenue, while losses are projected to narrow to $58 million from $64.7 million in the prior quarter.
- Another key catalyst comes next week at a medical summit, where ImmunityBio will present new Anktiva comparisons against rival bladder cancer therapies.
ImmunityBio, Inc. (IBRX) shareholders are headed for a major catalyst stretch that includes a key FDA filing decision on its flagship immunotherapy, Anktiva, quarterly earnings, and fresh cancer-treatment data next week.
IBRX stock ended 11% higher at $8.38 on Wednesday, logging its best session in over a month.
Anktiva FDA Filing Clock Expires Soon
The FDA’s standard 60-day review window for ImmunityBio’s resubmitted supplemental biologics license application (sBLA) for Anktiva expires Thursday. The company refiled the application on March 9, seeking approval for Anktiva plus BCG in patients with a difficult-to-treat form of non-muscle invasive bladder cancer with papillary tumors. During the 60-day period, the FDA determines whether the application is sufficiently complete to formally accept it for full review and to assign a target action date.
The filing is backed by long-term data from the Quilt 3.032 study, which ImmunityBio said showed that 58.2% of patients remained cancer-free after 12 months, while more than 80% avoided bladder removal surgery over three years.
IBRX Q1 Preview
Wall Street expects first quarter (Q1) revenue of $43.9 million, up 15% from the previous quarter, while losses before interest, taxes, depreciation and amortization (EBITDA) are projected to narrow to $58 million from $64.7 million in the prior quarter. Analysts also expect a loss of $0.07 per share.
The estimates align with ImmunityBio’s preliminary figures, in which the company reported expected Q1 net product revenue of $44.2 million, up 168% from the previous year and reflecting continued quarter-over-quarter growth since Anktiva’s commercial launch. The company also said it ended the quarter with about $381 million in cash, cash equivalents and marketable securities. Koyfin data also shows that analysts maintain an average 12-month price target of $15.75, implying an 88% upside from current levels.
Another key catalyst comes next week at the American Urological Association Annual Meeting in Washington, D.C., where ImmunityBio plans to present new Anktiva analyses comparing the therapy against rival bladder cancer treatments, including Johnson & Johnson’s TAR-200 and Nadofaragene Firadenovec. Founder Patrick Soon-Shiong is also expected to present new data around IL-15 immune activation and broader immunotherapy approaches.
FDA Leadership Turmoil Adds New Twist
The Anktiva decision is looming amid growing controversy around leadership inside the FDA’s biologics and cancer divisions. Vinay Prasad, the FDA official overseeing biologics, vaccines, and gene therapies, served his final day at the agency recently after facing backlash for several biotech drug rejections and accelerated-approval disputes.
The pressure has now shifted towards FDA Commissioner Marty Makary himself after criticism of the agency’s handling of cancer reviews and comments he made during a recent CNBC interview defending the FDA’s rejection of Replimune’s melanoma therapy, RP1. The controversy deepened further after Makary selected Katherine Szarama as the acting replacement for Prasad. Szarama has previously criticized accelerated approvals and clinical datasets lacking traditional control arms.
The turmoil has fueled speculation that pressure from U.S. President Donald Trump’s administration could eventually force broader changes at the FDA and push the agency toward a more flexible stance on accelerated approvals.
How Do Retail Traders Feel About IBRX?
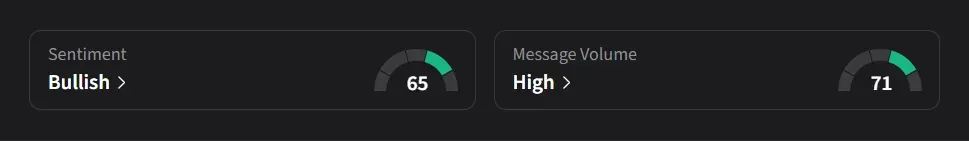
On Stocktwits, retail sentiment for IBRX was ‘bullish’ amid ‘high’ message volume.
One user said, “FDA is going to accept the sbla this week and issue a PDUFA date of 3 to 6 months. Most likely it'll be November 9th. Stock will be well over $10 by this Friday. Cheers.”
Another user said, “What makes ASCO & AUA Conf Trial news great is the FDA BLA acceptance comes before.”
IBRX stock has surged 317% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.














/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2213700319_jpg_af5569acdc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Snap_jpg_11a44b44a0.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2213700945_jpg_100e788722.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_immunitybio_jpg_eb6402d336.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Stock_chart_jpg_6c4cc95d17.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)