Advertisement|Remove ads.
IMVT Stock On Track For 4-Month Low After Phase 3 Setback In Thyroid Eye Disease Trials

- The studies aimed to show a meaningful reduction in eye bulging after 24 weeks of treatment.
- Patients experienced greater improvement in eye bulging during the initial 12-week high-dose treatment period than during the subsequent low-dose phase.
- Immunovant to focus on developing IMVT-1402 to treat multiple autoimmune conditions, including Graves’ disease.
Advertisement|Remove ads.
Shares of Immunovant (IMVT) fell 12% in pre-market trading on Thursday, after the biotech firm reported disappointing topline results from two late-stage trials to treat thyroid eye disease.
If the pre-market levels hold after the opening bell, IMVT shares would drop to their lowest levels in nearly four months.
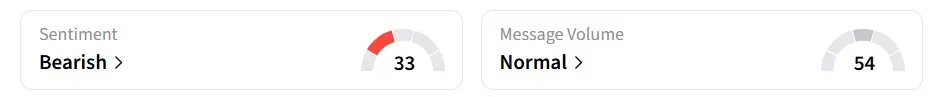
Retail sentiment on Stocktwits turned ‘bearish’ from ‘neutral’ a day earlier.
Advertisement|Remove ads.
Early Signals Hint At A Dosing Problem, Not A Dead Drug
Immunovant said topline results from two of its Phase 3 studies of batoclimab in adults with active, moderate-to-severe thyroid eye disease (TED) did not meet the primary endpoint. The studies aimed to show a meaningful reduction in eye bulging after 24 weeks of treatment, but the results fell short. However, the safety profile remained consistent with earlier findings, with no new safety concerns identified.
Despite missing the primary goal, the data offered some encouraging signals. Patients experienced greater improvement in eye bulging during the initial 12-week high-dose treatment period than during the subsequent low-dose phase. This suggests that deeper suppression of Immunoglobulin G (IgG) antibodies may be key to achieving better outcomes, the company noted.
Advertisement|Remove ads.
Hyperthyroid patients in the trials achieved thyroid hormone normalization rates similar to those observed in earlier Phase 2 studies in patients with Graves’ disease, an immune system disorder that affects the thyroid gland.
Focus On IMVT-1402
Following these results, Immunovant is shifting its focus to advancing IMVT-1402, a next-generation therapy under development across multiple autoimmune conditions, with Graves’ disease as a key priority. Early Phase 2 data suggest this approach could help modify the disease, and topline results from late-stage studies are expected in 2027.
The TED Drug Race Heats Up
Earlier this week, Viridian Therapeutics (VRDN) shares crashed after the company reported its Phase 3 results for Elegrobart in thyroid eye disease. While the study met its primary goal, showing strong reductions in eye bulging compared with placebo, improvements in inflammation were limited, and there were also reports of tinnitus.
Advertisement|Remove ads.
IMVT shares have declined around 2% so far in 2026.
Read also: Why Did MOBX Stock Crash 25% In Pre-Market Today?
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Advertisement|Remove ads.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_Trade_desk_logo_resized_c0229eb2ab.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2255802445_jpg_427b92fbb5.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_fuelcellenergy_resized_ef1a57cfe6.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_oscar_health_jpg_89aa74c6fe.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2217651425_jpg_0d7dc8a6cd.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spy_jpg_a85fe2a8bc.webp)