Advertisement|Remove ads.
SLS Stock Pops After-Hours As Loss Shrinks, Cash Swells — All Eyes On High Stakes AML Trial In 'Pivotal' 2026
- Net loss in FY25 narrowed to $26.9 million from $30.9 million a year ago, with R&D costs falling to $16 million.
- Phase 3 Regal AML trial nears final analysis, with 72 of 80 required survival events reached, keeping focus on upcoming topline data.
- SLS009 advances into first-line AML trials, backed by prior 46% response rate data and supported by upcoming AACR preclinical results.
Shares of Sellas Life Sciences Group Inc. (SLS) posted modest gains in extended trading on Thursday after the company reported a narrower full-year loss and a stronger cash position, as investors turned their focus to a closely watched Phase 3 AML trial nearing its final trigger.
SLS stock snapped three straight sessions of losses on Thursday to end 5% higher at $5.20, and gained 0.4% in the extended session. However, shares are headed for their worst week in over a month.
SLS FY25 Review
The biotech firm reported a net loss of $26.9 million, or $0.25 per share, for the year ended Dec. 31, 2025, compared with a net loss of $30.9 million, or $0.5 per share, in 2024. Research and development expenses declined to $16 million from $19.1 million, while general and administrative expenses were largely unchanged at $12.3 million.
The decline in expenses was primarily driven by lower clinical trial and regulatory consulting costs following the completion of enrollment in the Phase 3 Regal study in early 2024. Sellas also reported receiving $67.2 million in gross proceeds from warrant exercises during 2025.
Sellas ended 2025 with $71.8 million in cash and cash equivalents and said it received an additional $42.6 million in proceeds from warrant exercises in the first quarter of 2026.
“With a catalyst rich outlook, continued clinical execution, our strongest financial position in history, and expansion into earlier treatment settings, we look forward to an important year ahead,” CEO Angelos Stergiou said in a statement.
Sellas said 2026 is shaping up to be a “pivotal year,” citing progress across its acute myeloid leukemia (AML) programs and upcoming catalysts.
GPS Trial Moves Closer To Final Analysis
Investor attention remains centered on Sellas’ Phase 3 Regal trial evaluating galinpepimut-S (GPS) in AML patients who have achieved remission following second-line salvage therapy.
The event-driven trial is designed to trigger final analysis upon reaching 80 survival events. The company said 72 events had been recorded as of Dec. 26, with the study remaining fully blinded. Once the required event threshold is reached, Sellas will proceed with database lock, statistical analysis, unblinding and disclosure of topline results.
Sellas Builds Momentum With SLS009 Pipeline
The company also highlighted its second pipeline candidate, SLS009, which is being advanced into earlier lines of treatment, with the first patient already dosed in an 80-patient Phase 2 trial in newly diagnosed AML.
In January, Sellas entered a collaboration with Impact-AML to advance the program in Europe, with a study expected to enroll around 40 patients starting in the second quarter.
The program builds on previously reported Phase 2 data, where SLS009, used alongside azacitidine and venetoclax, two commonly used AML therapies, achieved a 46% response rate in relapsed or hard-to-treat AML, including 29% complete responses, with improved survival and no dose-limiting toxicities observed.
Sellas Flags ‘Compelling’ Early Data For SLS009
The company also pointed to preclinical findings supporting SLS009, with data to be presented at the American Association for Cancer Research (AACR) next month, showing the drug induces cancer cell death in AML models, including those with high-risk ASXL1 and TP53 mutations, with early changes seen in key survival proteins such as MCL-1 and survivin.
The presentation comes ahead of earlier preclinical results shared at the European Society for Medical Oncology Congress, where SLS009 showed a statistically significant survival benefit in a T-cell leukemia model, both as a standalone therapy and in combination with venetoclax.
Stergiou called the findings “compelling new data,” adding that SLS009 showed activity in AML models with high resistance.
How Did Stocktwits Users React?
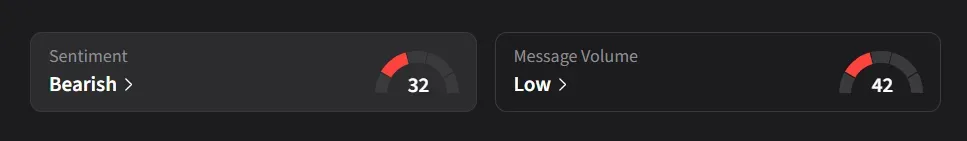
On Stocktwits, retail sentiment for SLS was ‘bearish’ amid ‘low’ message volume.
One user said the stock’s direction largely depends on the upcoming GPS data, with strong results potentially driving sharp gains and weaker outcomes posing downside risk.
Another user said, “THE DATA JUST NEED TO BE “GOOD ENOUGH” (HR 0.636) TO GET FDA APPROVAL. SO IT IS EITHER $70 OR $180.”
SLS stock has rocketed over 300% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_marvell_technology_logo_resized_jpg_be0d6f32f6.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_strategy_michael_saylor_jpg_edc8d048a9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2247045310_jpg_c3c3feb79b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_spacex_jpg_7d337cbe8a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_verastem_jpg_8ed70d9d9a.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2253972629_jpg_f4ce2c8efa.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)