Why Did CANF Stock Soar 25% Today?

- The study enrolled 20 high-risk patients with advanced, metastatic disease who had received one or more prior therapies.
- The company said that the secondary endpoints included overall and progression-free survival.
- Can-Fite announced exercise of certain outstanding warrants to purchase up to 795,869 ADS at a reduced price of $5 per share.
Shares of Can-Fite BioPharma (CANF) surged more than 40% in pre-market trading on Wednesday after the company reported positive results from its Phase 2A open-label study of namodenoson in patients with advanced pancreatic ductal adenocarcinoma (PDAC) who had previously failed other treatments.
The trial met its primary safety endpoint, showing that the drug was very well tolerated, with no new safety concerns observed. The study enrolled 20 high-risk patients with advanced, metastatic disease who had received one or more prior therapies.
The company said that the secondary endpoints included overall and progression-free survival, with follow-up ongoing and one-third of patients still alive at data cut-off, as more mature results are expected to be presented at upcoming scientific meetings.
Namodenoson is a selective A3 adenosine receptor agonist that has shown anti-tumor activity in preclinical models and is also being studied in advanced liver cancer. The drug has received Orphan Drug Designation from the U.S. Food and Drug Administration (FDA) for the treatment of pancreatic cancer.
Exercising of Warrants for $4.0 Million Proceeds
Can-Fite BioPharma also announced a definitive agreement for the immediate exercise of certain outstanding warrants to purchase up to 795,869 American Depositary Shares at a reduced price of $5 per share, down from $9.34. The transaction is expected to close around March 5, 2026.
In connection with the cash exercise, the company will issue new unregistered warrants to purchase up to 1,591,738 ADSs at $5 per share, exercisable for 24 months.
Can-Fite expects gross proceeds of about $4.0 million to support research, clinical trials, and general corporate purposes.
How Did Stocktwits Users React?
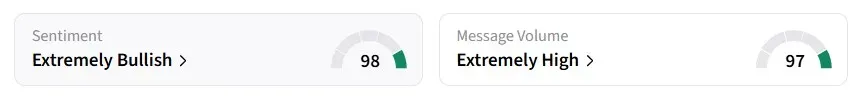
Retail sentiment for CANF on Stocktwits turned 'extremely bullish' from 'bullish' a day earlier, amid 'extremely high' message volumes.
One user said the company is a "great buyout candidate."
Year-to-date, the stock gained 63%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)