Advertisement|Remove ads.
ERAS Stock Slumps Nearly 40% After-Hours: Fine Print In 'Positive' Cancer Trial Reveals Treatment-Related Death

- The death followed Grade 3 pneumonitis in a 66-year-old pancreatic cancer patient receiving a 24 mg dose.
- ERAS-0015 showed strong early tumor response rates, including 62% in KRAS-mutant lung cancer.
- Additional data from the Auroras-1 trial are expected in 2027, while results from its other candidate, ERAS-4001, are expected later this year.
Advertisement|Remove ads.
Shares of Erasca, Inc. (ERAS) plunged nearly 40% in extended trading on Monday after investors spotted a treatment-related death buried within safety disclosures, even as the company flagged strong early tumor response rates from a Phase 1 study of its experimental cancer therapy ERAS-0015.
ERAS stock slid nearly 11% on Monday, logging its worst session in nearly eight months.
Treatment-Related Death Surfaces In ERAS Phase 1 Trial
According to a fresh filing, one patient experienced Grade 3 pneumonitis that progressed to a treatment-related death. The patient was a 66-year-old male with metastatic pancreatic cancer receiving a 24 mg dose of ERAS-0015 who had pulmonary metastases and prior lung procedures before entering the study.
Advertisement|Remove ads.
The patient later discontinued treatment after developing pneumonitis one month after therapy initiation and later died following withdrawal of supportive care.
ERAS Cancer Trial Shows Strong Early Cancer Responses
Alongside the safety update, Erasca reported strong early response signals from the company’s Auroras-1 study across KRAS-mutant cancers. In previously treated non-small cell lung cancer, ERAS-0015 showed a 62% tumor response rate at eight weeks, rising to 75% among patients who had already received immunotherapy and platinum chemotherapy. In pancreatic cancer, the therapy showed a 40% response rate at fourteen weeks, with most responding patients still on treatment at the time of analysis.
The company also reported that all 14 evaluable patients showed large reductions in circulating tumor DNA levels. Based on these results, Erasca selected 24 mg and 32 mg once daily as the doses to move forward into expansion studies.
Advertisement|Remove ads.
Looking ahead, the company expects additional monotherapy expansion and combination dose-escalation data from the Auroras-1 study in the first half of 2027. Preliminary Phase 1 data for ERAS-4001, its pan-KRAS inhibitor program, is expected in the second half of this year.
RevMed Patent Claims Add Pressure
Separately, Erasca disclosed in the new filing that it received a letter from the legal counsel for Revolution Medicines alleging that ERAS-0015 may infringe compositions claimed in a U.S. patent under the doctrine of equivalents. The letter also alleged possible trade-secret misappropriation from a third-party patent relating to ERAS-0015 and challenged comparisons between ERAS-0015 and RMC-6236 made in public disclosures.
RMC-6236 is a cancer drug being developed by Revolution Medicines to treat tumors driven by KRAS mutations. Unlike older targeted therapies that focus on a single mutation, it works across multiple KRAS mutation types, which are common in cancers such as lung and pancreatic cancer. It is still in early-stage clinical testing.
Advertisement|Remove ads.
Revolution Medicines requested that Erasca cease manufacturing, using or selling ERAS-0015 outside Hatch-Waxman safe-harbor protections and stop making “untrue comparative statements.” However, Erasca said it believes the allegations are without merit and intends to contest them vigorously.
How Do Retail Traders Feel About ERAS?
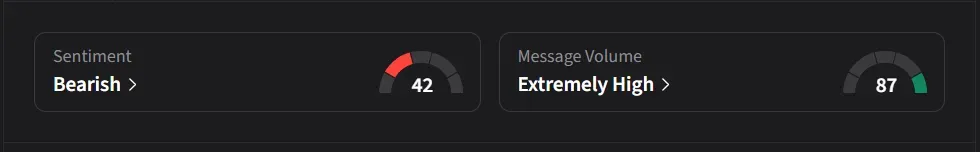
On Stocktwits, retail sentiment for ERAS slipped to ‘bearish’ from ‘neutral’ levels from a day ago amid ‘extremely high’ message volume.
One user said, “Long-term effert on stock price depends on Erasca's relationship with FDA and whether a clinical hold is issued. Honesty and timeliness is paramount in FDA interactions.”
Advertisement|Remove ads.
Another user said, “The Reality Check: If a drug makes a patient "decide" to stop all care, the toxicity is the problem, not the patient. You aren't buying a dip; you're buying a bio-hazard.”
Advertisement|Remove ads.
ERAS stock has surged nearly 1,300% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: Lucid Stock Is Crashing Hard And Leaves Retail Wondering If Saudi PIF Buyout Is A Good Exit Strategy
Advertisement|Remove ads.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263602546_jpg_a0f4e7e3ca.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2071907975_jpg_85e059f13e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/unnamed_jpg_9dff551b50.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2200882557_jpg_53f3e467bc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2238077141_jpg_984e46421c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_nyse_OG_2_jpg_0e4e1c68f2.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_sirius_xm_logo_resized_8dc3ebc68d.jpg)