Advertisement|Remove ads.
IBRX Stock Jumps After-Hours: Bulls Cheer ‘Unstoppable’ Global Expansion Amid US Regulatory Heat For Anktiva

- ImmunityBio plans to present new Anktiva bladder-cancer data at the upcoming AUA meeting.
- The company said it will also provide updates on recombinant BCG research and efforts to improve BCG access.
- Retail traders had already fueled speculation around ImmunityBio’s overseas expansion plans after tracking Soon-Shiong’s private jet in Japan.
Shares of ImmunityBio, Inc. (IBRX) jumped about 2% in extended trading on Tuesday after founder Patrick Soon-Shiong touted “unstoppable” global expansion for Anktiva, boosting investor optimism around the cancer therapy’s international footprint despite ongoing FDA scrutiny in the U.S.
IBRX stock had also jumped over 2% in Tuesday’s regular session, logging its fourth straight session of gains.
IBRX Eyes Bigger Bladder Cancer Market
Late Tuesday, Soon-Shiong signaled confidence in ImmunityBio’s international push, posting on X: “Global expansion. Unstoppable.” The comments came shortly after ImmunityBio announced plans to present new comparative data for Anktiva at the upcoming American Urological Association Annual Meeting. The company said it will present data comparing Anktiva used with standard BCG therapy against rival bladder-cancer treatments Nadofaragene firadenovec-vncg and Johnson & Johnson’s TAR-200 in certain patients with early-stage bladder cancer, including those with papillary disease, where the cancer has not spread into the muscle layer.
ImmunityBio said the presentations will provide additional information on efficacy, durability and safety in the absence of direct head-to-head studies. The company will also share updates on its recombinant BCG research in patients who have not previously received BCG treatment, along with efforts to improve access to BCG.
Japan Buzz And Global Expansion
Soon-Shiong’s comments also added fuel to growing retail speculation surrounding ImmunityBio’s overseas expansion strategy, particularly after traders spent the past week tracking the founder’s private jet movements in Japan using public flight-transponder data.
Some investors speculated that an extended Japan visit pointed at potential partnership talks or future commercialization plans, although the company has not announced any such agreements. Retail traders have increasingly viewed Japan as a potential growth market for Anktiva following recent approvals and launches across Europe, Macau and Saudi Arabia.
The company’s global expansion strategy has also gained momentum alongside progress at its Dunkirk, New York manufacturing facility. Last week, CEO Richard Adcock called the long-delayed site “all systems go” after local officials backed progress on the facility, which is expected to become a key production hub for Anktiva.
Adcock had also highlighted the pace of Anktiva’s international growth, saying the therapy had expanded from approvals in “zero countries” to 34 countries and territories since the company began building out its commercialization strategy.
Anktiva FDA Scrutiny Remains In Focus
Despite growing overseas plans, Anktiva continues facing regulatory pressure in the U.S. Last year, the FDA issued a Refusal to File (RTF) letter for ImmunityBio’s supplemental application seeking expanded use of Anktiva in papillary disease. The company later resubmitted the filing with updated efficacy data and longer-term follow-up.
The therapy has also faced scrutiny for promotional claims, with the FDA issuing warning letters regarding statements tied to broader cancer applications. Soon-Shiong publicly pushed back on parts of the agency’s criticism. Investor focus on the FDA intensified again this week after a Wall Street Journal opinion piece criticized FDA Commissioner Marty Makary’s role and highlighted internal tensions surrounding cancer and rare-disease drug reviews.
Anktiva is approved alongside standard BCG therapy for certain bladder-cancer patients who no longer respond to treatment and is also being studied across lung cancer, pancreatic cancer, colorectal cancer, glioblastoma and immune-related conditions including long COVID and sepsis.
How Do Retail Traders Feel About IBRX?
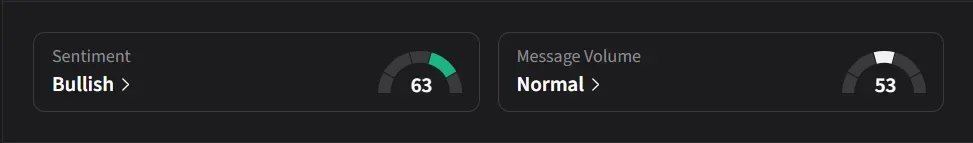
On Stocktwits, retail sentiment for IBRX jumped to ‘bullish’ from ‘neutral’ levels over the past day amid a 235% surge in 24-hour message volumes.
One user tracking Soon-Shiong’s private jet movements speculated about potential Europe expansion plans, while another trader said, “never saw this route before if Europe/UK usually turns north early on.”
Another trader said, “Dr. Pat is a genius. He knew the only way to defeat this country's corruption was to go to the world where some countries do not hose their citizens in favor of big money lobbyists.”
IBRX stock has surged 261% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: AMD Stock Soars On Q1 Data Center Revenue Surge, Upbeat Q2 Guidance

/filters:format(webp)https://news.stocktwits-cdn.com/large_Abercrombie_resized_jpg_320d8a63ad.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_TSMC_jpg_616893a30b.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Zscaler_jpg_c6a5978bfc.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263602541_1_jpg_988d7f324e.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_Groupon_logo_resized_jpg_9aa7f77f8c.webp)