Advertisement|Remove ads.
IBRX Stock Eyes 2-Week Winning Streak: Traders Say Founder’s Quiet Japan Trip Signals ‘Massive Deal' In The Works

- Some traders outlined a hypothetical $10 billion Japan stake scenario, suggesting a partner could take a 10% position for future supply commitments.
- Retail investors see Japan as a logical next expansion step following approvals and commercialization progress across the U.S., U.K., European Union, Macau and Saudi Arabia.
- Japan’s Conditional Early Approval program drew attention from traders, who noted the possibility of earlier reimbursement-based commercialization timelines.
Advertisement|Remove ads.
Shares of ImmunityBio, Inc. (IBRX) are headed for a second straight week of gains as retail traders speculated that an unusually long and quiet Japan trip by founder Patrick Soon-Shiong could signal a potential deal as part of the cancer drugmaker’s global expansion plans.
IBRX stock has jumped over 6% so far this week after climbing over 1% on Thursday alone amid the Japan deal speculations. Shares have surged by more than 200% over the last year.
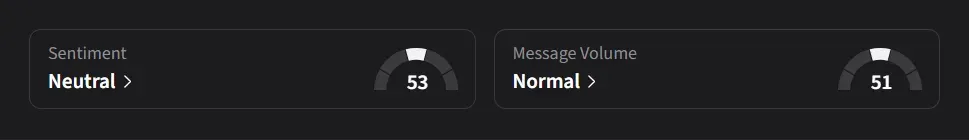
Retail sentiment on Stocktwits for IBRX has largely been in the ‘neutral’ to ‘bullish’ and ‘extremely bullish’ range over the past six months amid nearly a 250% surge in yearly message volumes.
Advertisement|Remove ads.
Retail Tracks CEO Japan Trip For Deal Signs
Retail investors tracking Patrick Soon-Shiong’s private Gulfstream G650 jet using publicly available aircraft transponder data on platforms such as JetSpy said the length of the Tokyo visit stood out compared with previous overseas trips. However, the company has not announced any agreements regarding a Japan deal.
“Flight logs showing this being longest trip for single country I have seen yet,” one trader said, adding the lack of updates compared with earlier trips to Saudi Arabia, the UAE and Portugal suggested “a massive deal” in the works.
Some traders speculated that the founder’s travel plans in Japan was for potential industry meetings.
Advertisement|Remove ads.
“Recently left Tokyo for Kobe, Japan. The bio hub of the country,” another user said. Meanwhile, a third user said, “Dr. Pat is not in Japan for a vacation but for business. Longs expect super news in the next 3 days.”
Traders Float Japan Stake Deal Scenario
Some traders went further, outlining potential partnership or equity-investment scenarios. The user sees a hypothetical scenario in which a Japanese partner could acquire a 10% stake in ImmunityBio through a $10 billion tender-style investment for future supply commitments for its immunotherapy platform, Anktiva. They suggested that such a deal could provide upfront capital to support commercialization in Japan.
Others suggested that Japan could be the next logical step in the company’s international rollout after recent approvals across regions including the U.S, U.K, the European Union, Macau and Saudi Arabia.
Advertisement|Remove ads.
“This disciplined expansion into new markets, with Japan and China clearly in the crosshairs, is building the momentum necessary to make the stock price skyrocket as the company evolves into a true global powerhouse,” the user said.
Notably, Soon-Shiong recently reposted a post from MAHA Action, health-policy advocacy group, on X warning that the U.S. risks falling behind China in drug approvals and clinical-trial activity. The post said: "We are losing scientists, we’re losing our IPs, we’re losing the best researchers.”
Japan Early Approval Program Draws Retail Focus
Retail traders also highlighted Japan’s Conditional Early Approval program as a possible driver of earlier commercialization timelines if the company pursues regulatory engagement there. One trader flagged that “once a drug receives Conditional Early Approval in Japan, it is eligible for National Health Insurance reimbursement.”
Advertisement|Remove ads.
“This effectively turns the Japanese population of 125 million into a revenue-generating Phase 3 trial,” the user said. Japan’s health-policy efforts to address delays in access to innovative medicines have also been cited by them as supportive of overseas immunotherapy entrants.
FDA Scrutiny Persists Even Amid Global Approvals
Retail optimism about Japan also comes as Anktiva remains under heavy scrutiny from the U.S. Food and Drug Administration (FDA), which has flagged promotional claims tied to its approved use, even as the therapy continues to gain regulatory traction overseas.
Last month, regulators in Macau authorized the therapy for certain bladder-cancer patients, marking its first approval in Asia, while earlier this week it became commercially available in Saudi Arabia for bladder-cancer and non-small-cell lung-cancer indications following regional distribution rollout.
Advertisement|Remove ads.
Traders have previously expressed optimism that U.S. President Donald Trump could again support international momentum for anktiva, after the founder said earlier that discussions with the president helped advance the therapy’s rollout in the Middle East.
Anktiva received approval in 2024 with standard BCG for certain bladder-cancer patients who no longer respond to treatment. It is now being studied in non-small cell lung cancer, pancreatic cancer, colorectal cancer, glioblastoma, and immune-related conditions, including severe pneumonia, sepsis, long COVID, and acute respiratory distress syndrome.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Advertisement|Remove ads.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2233660811_jpg_d8338bcfaf.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2209880858_jpg_f9932067ca.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Aashika_Suresh_Profile_Picture_jpg_2acd6f446c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_809251022_jpg_fc0a213f4e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2268516558_jpg_8245ea1920.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_oil_stocks_jpg_6935abe231.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2279256317_jpg_7d0ae1fc5e.webp)