Advertisement|Remove ads.
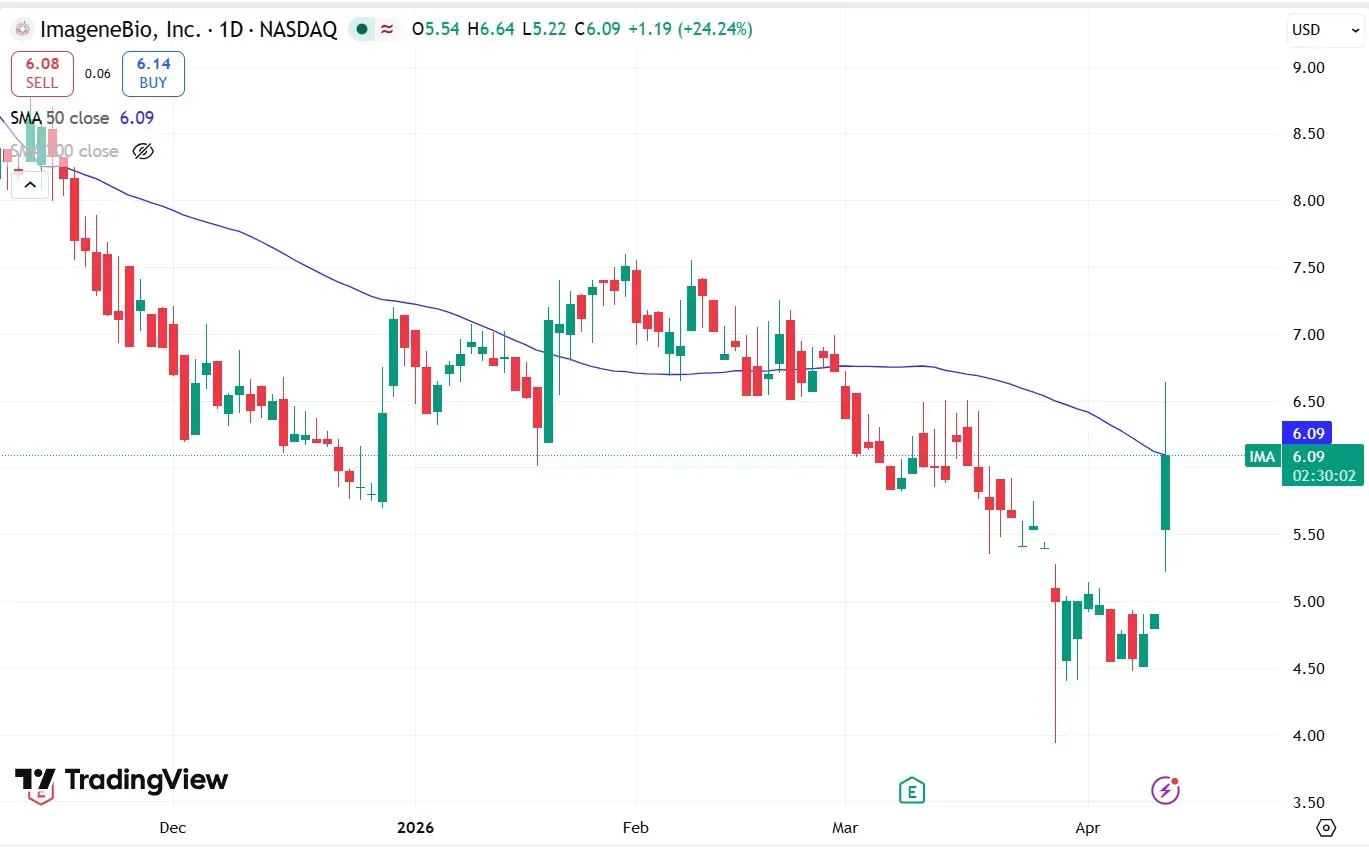
IMA Stock Surges 24%, Breaks 50-DMA For First Time Since February – What's Driving The Rally?

- ImageneBio said it plans to use the proceeds to support the development of IMG-007 for alopecia areata and late-stage work in atopic dermatitis.
- The biotech firm is currently conducting its Phase 2b ADAPTIVE trial for IMG-007, with topline results expected in 2027.
- ImageneBio will present the latest data from the study at the Annual Society for Investigative Dermatology Meeting in May.
Shares of ImageneBio, Inc. (IMA) surged more than 24% on Monday, crossing their 50-day moving average (50-DMA) for the first time since Feb. 27, 2026, after the biotech firm announced fresh funding from institutional investors in the form of a $30 million private placement.
Source: TradingView
ImageneBio said it plans to use the proceeds to support the development of IMG-007 for alopecia areata and late-stage work in atopic dermatitis. The deal involves the sale of pre-funded warrants to purchase over 5.7 million shares at $5.199 per share.
The financing round is led by new investor Coastlands Capital, with participation from Trails Edge Capital Partners and existing backers Omega Funds and OrbiMed.
What’s The Latest On Drug To Treat Dermatitis?
ImageneBio is currently conducting a Phase 2b ADAPTIVE trial for IMG-007, with topline results expected in 2027. The study, which began enrollment in mid-2025 across North America, continues to show a favorable safety and tolerability profile, as indicated by a recent blinded review. A Blinded review means the people running a clinical trial do not know which patients are receiving the treatment.
IMG-007 has been well tolerated across studies involving more than 150 patients, with no serious safety concerns reported. Reported side effects have been minimal, with very low rates of mild injection-site reactions.
The company has also recently submitted a protocol update to expand dosing strategies and explore more patient-friendly treatment schedules. ImageneBio will present the latest data from the study at the Annual Society for Investigative Dermatology Meeting in May.
How Did Retail Traders React?
Retail sentiment for IMA on Stocktwits turned ‘extremely bullish’ from ‘bullish’ a day earlier, amid ‘extremely high’ message volumes.
One bullish user said buying the stock was a “no-brainer,” comparing the company’s market cap to the funding amount.
Another user expects the stock to climb all week.
Year-to-date, the stock has gained around 5%.
Read also: CAG Stock Crashes To Near 17-Year Lows, But Retail Sees ‘Buying Opportunity Of A Decade’
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)