Advertisement|Remove ads.
Novo Nordisk Receives FDA Approval For Higher Dose Of Its Weight-Loss Drug Wegovy
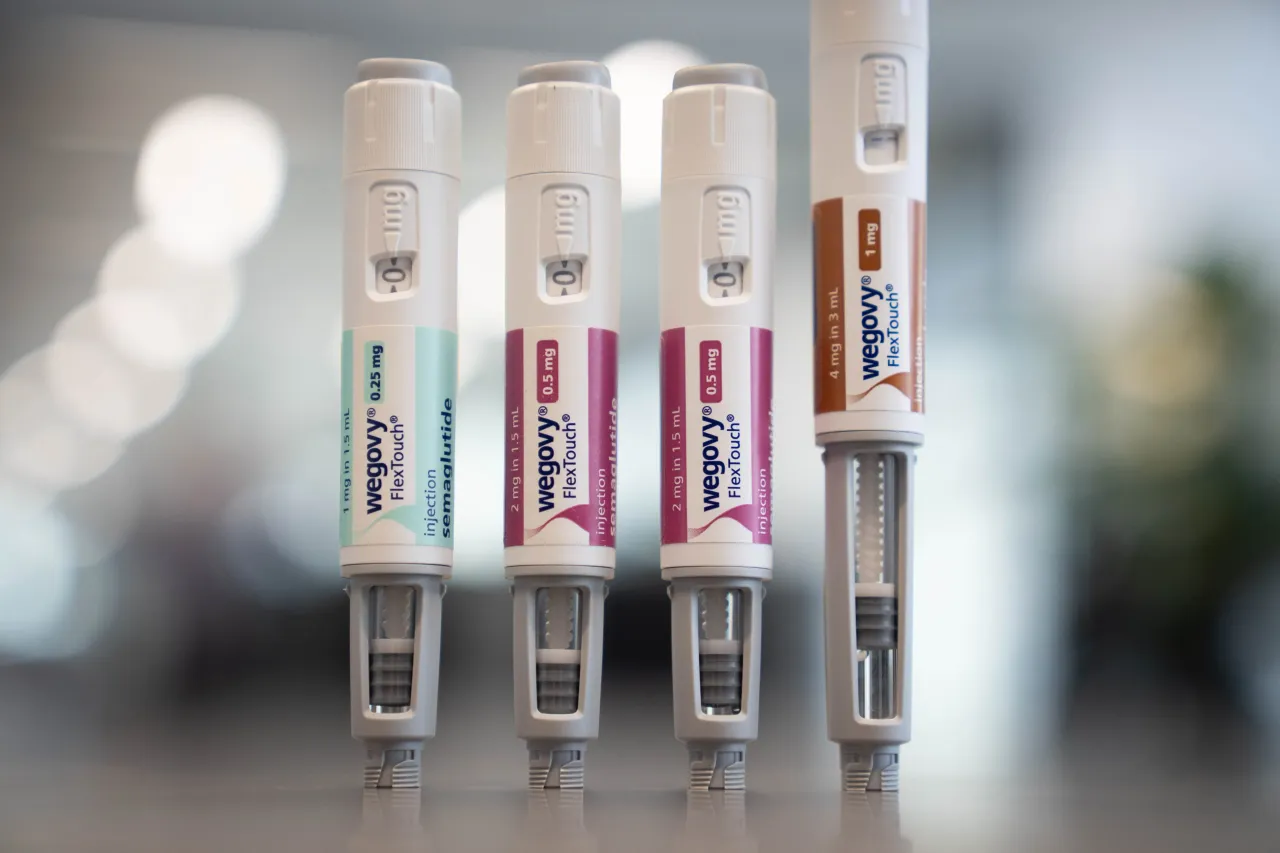
- Novo Nordisk states that Wegovy HD in a single-dose pen is expected to be available in April 2026.
- Wegovy injectable was available in weekly doses of 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg until now.
- Novo Nordisk added that Wegovy 7.2 mg has already been approved in the U.K. and the European Union as three 2.4 mg doses per week.
Novo Nordisk AS (NVO) on Thursday said the U.S. Food and Drug Administration (FDA) has granted accelerated approval for a higher-dose version of its blockbuster weight-loss drug Wegovy, expanding options for patients seeking greater weight reduction.
The company stated that Wegovy HD, a once-weekly injectable semaglutide at 7.2 mg, has received FDA accelerated approval based on results from the STEP UP trial program.
Novo Nordisk shares were down nearly 2% in Thursday morning’s trade. Retail sentiment on Stocktwits around the company trended in the ‘bearish’ territory at the time of writing.
When Will Wegovy HD Be Available?
Novo Nordisk stated that Wegovy HD in a single-dose pen is expected to be available in April 2026. Wegovy injectable was available in weekly doses of 0.25 mg, 0.5 mg, 1 mg, 1.7 mg, and 2.4 mg until now.
“The FDA awarded a Commissioner’s National Priority Voucher for Wegovy HD, accelerating its review and underscoring its potential to address critical patient needs and national health priorities in the U.S.,” the company stated.
Novo Nordisk added that Wegovy 7.2 mg has already been approved in the U.K. and the European Union as three 2.4 mg doses per week. It added that the regulatory decisions in the two markets for the single-dose pen are expected in the second half of 2026.
STEP UP Trial Shows Stronger Weight Loss
Novo Nordisk stated that Wegovy HD’s accelerated approval by the FDA was based on a 72-week study that showed an average weight loss of about 20.7% in patients.
The company added that nearly one in three patients receiving Wegovy HD experienced a 25% or greater weight loss.
In the STEP UP type 2 diabetes trial, the company said participants demonstrated a mean weight loss of 14.1%, while about one in five patients experienced a weight loss of 20% or more.
Intensifying Competition
Novo Nordisk’s launch of Wegovy HD follows CEO Mike Doustdar's warning of pricing headwinds in 2026 amid an increasingly competitive market.
The Dutch company expects its 2026 sales to decline between 5% and 13% in constant-currency terms.
“However, we are very encouraged by the promising early uptake from the U.S. launch of Wegovy pill, and we remain confident in our ability to drive volume growth over the coming years,” Doustdar added.
Novo Nordisk rival Eli Lilly & Co. (LLY) expects fiscal year 2026 revenue to rise between 23% and 28%, highlighting a divergence in the outlooks of the two companies this year.
NVO stock is down 28% year-to-date and 54% over the past 12 months. The VanEck Pharmaceutical ETF (PPH) is up 9% over the past 12 months, while the Putnam Focused Large Cap Value ETF (PVAL) is up 20%.
Also See: American Rebel Shares Sink More Than 40% — What's Driving The Plunge?
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1233265488_jpg_60af97169a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275851163_jpg_b115c861be.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2237640344_jpg_bc97a7240c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2227347116_jpg_6a738ad597.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2201127733_jpg_293396ee32.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269243885_jpg_4cc426aadd.webp)