Advertisement|Remove ads.
VKTX Stock Slips Premarket: A Surprise Weight Loss Drug Beats Novo Nordisk In Early Animal Tests

- The company's obesity drug candidate VK3019 showed stronger early weight-loss signals than Novo Nordisk’s amylin therapy.
- The company said enrollment has been completed across both Phase 3 Vanquish trials evaluating weekly injectable VK2735.
- Viking plans to launch Phase 3 development of the oral VK2735 formulation in Q4.
Shares of Viking Therapeutics, Inc. (VKTX) slipped 0.4% in premarket trading on Thursday even after the drugmaker said that its newer obesity drug candidate delivered stronger early weight-loss signals than Novo Nordisk’s comparable therapy in animal studies.
VKTX stock is on track to post its second straight month of losses, sliding nearly 4% so far in April.
VK3019 Boosts Viking’s Challenge To Novo Nordisk
Investor sentiment has largely turned on optimism toward VK3019, Viking’s obesity candidate targeting the amylin receptor, a mechanism increasingly viewed as complementary to GLP-1-based therapies.
CEO Brian Lian said on the company’s recent earnings call that early testing suggested the compound compared favorably with Novo Nordisk’s amylin candidate: “When we looked at data in rodents, it seemed to be better than Cagrilintide.”
He also noted stronger signals relative to Viking’s own lead obesity therapy in animal testing, saying, “When we looked in obese monkeys, it seemed to provide better weight loss than VK2735.”
“it was very potent on the receptors, very good PK [pharmacokinetics] profile that would be amenable to weekly dosing, we think, and that marries up nicely with the 2735-PK profile.” The company recently filed an investigational new drug application for VK3019 and expects to begin Phase 1 testing this quarter.
Viking Eyes Combo Obesity Strategy
Viking also confirmed early discussions around combination development linking VK3019 with VK2735, pointing to a broader multi-mechanism obesity platform strategy. Lian said that the amylin program may expand treatment flexibility for patients unable to tolerate GLP-1 therapies.
“Nice opportunity with this mechanism to potentially target people who are a little lower BMI… or people who maybe can’t tolerate the GLP-1 and want to try something different,” Lian added.
Phase 3 Momentum Builds For Lead Candidate
While VK3019 drew attention for its early signals, Viking’s primary focus still remains VK2735, its dual GLP-1 and GIP receptor agonist being developed in both injectable and oral forms. The company said that enrollment has now been completed across both Phase 3 Vanquish trials evaluating weekly injectable VK2735 in adults with obesity and those with obesity plus type 2 diabetes.
Alongside the injectable program, Viking is preparing to advance its oral VK2735 tablet formulation into Phase 3 development later this year following encouraging mid-stage trial results. The company said, “Every patient’s weight loss journey is unique, and we consistently hear from health care providers about the need for flexible treatment and administration options.”
The company expects to initiate the Phase 3 oral program in the fourth quarter.
VKTX Q1 Review
Viking reported a first-quarter net loss of $158.3 million, or $1.37 per share, wider than the consensus estimate of a $0.91 per-share loss and more than triple the $45.6 million, or $0.41 per share, loss recorded a year ago.
The increase was largely driven by a sharp rise in research and development spending to advance its Phase 3 obesity trials and manufacturing activities. Research and development expenses rose to $150 million during the quarter from $41 million a year earlier.
Despite the higher spending levels, the company ended the quarter with approximately $603 million in cash, cash equivalents and short-term investments, compared with $706 million at the end of 2025.
How Do Retail Traders View VKTX?
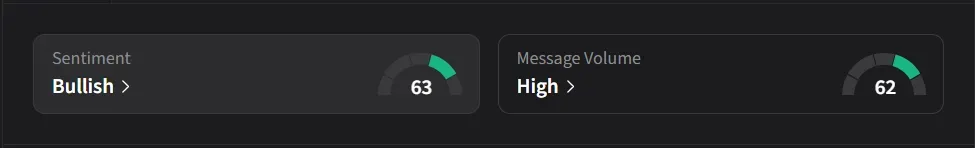
On Stocktwits, retail sentiment for VKTX jumped to ‘bullish’ from ‘bearish’ levels a day ago amid nearly a 200% jump in 24-hour message volumes.
One user said, “Viking therapeutics is synonymous with the word goldmine. I can't wait to see this cross the finish line. It's going to be a glorious day!”
Another user said VKTX was “BASICALLY Best in Class in everything.”
VKTX stock has risen 12% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_teladoc_logo_resized_f3ec80cc27.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Chinmay_Rautmare_Photo_jpeg_96dd37efd0.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2258806047_jpg_085e5f0fe0.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1233265488_jpg_60af97169a.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2214351115_jpg_9770916730.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Trending_stock_resized_may_jpg_bc23339ae7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263085688_1_jpg_87213df74e.webp)