Advertisement|Remove ads.
IBRX Stock Reverses Pre-Market Gains After ANKTIVA Combo Shows Stronger Bladder Cancer Responses – Retail Sees Consolidation Before Next Leg Up

- The study enrolled 366 patients, who were randomized to receive either BCG alone or ANKTIVA in combination with BCG.
- Interim analysis of the Phase 2 trial showed 84% of patients on the combo maintained a complete response, compared to 52% on BCG alone at nine months.
- ImmunityBio’s 431% revenue growth in Q4 was largely driven by ANKTIVA sales, the company said.
Advertisement|Remove ads.
Shares of ImmunityBio (IBRX) reversed pre-market gains to trade 3% lower on Thursday after the company completed enrollment ahead of schedule in its Phase 2 ANKTIVA trial and reported that an FDA-requested interim analysis showed significantly longer complete responses with no major safety concerns.
The Phase 2 trial studied ANKTIVA plus Bacillus Calmette-Guérin (BCG) in patients with BCG-naïve non-muscle invasive bladder cancer. The study enrolled 366 patients, who were randomized to receive either BCG alone or ANKTIVA in combination with BCG.
The company plans to submit a biologics license application by the fourth quarter of 2026. Last week, the European Commission granted conditional marketing approval for Anktiva to treat BCG-unresponsive non-muscle invasive bladder cancer with carcinoma in situ.
Advertisement|Remove ads.
ANKTIVA-BCG Combo Significantly Improves Complete Response Duration
An interim analysis requested by the U.S. Food and Drug Administration (FDA) showed that the combination significantly improved the duration of complete response, with no major safety concerns. At nine months, 84% of patients on the combo maintained a complete response, compared with 52% on BCG alone.
“We are encouraged by these interim results and await the final unblinding of the completed trial. If approved, ANKTIVA plus BCG could offer a new treatment option earlier in the disease course for patients with NMIBC CIS, building on the therapy’s existing approval in the BCG-unresponsive setting,” said Patrick Soon-Shiong, Chairman and Global Chief Scientific and Medical Officer of ImmunityBio.
ANKTIVA continues to power ImmunityBio’s growth, with fourth-quarter net product revenue soaring 431% to $38.3 million, beating Wall Street expectations on strong sales of the drug.
Advertisement|Remove ads.
How Did Stocktwits Users React?
Retail sentiment on for IBRX Stocktwits has remained ‘extremely bullish’ zone for the past six months, while message volumes have been ‘extremely high’.
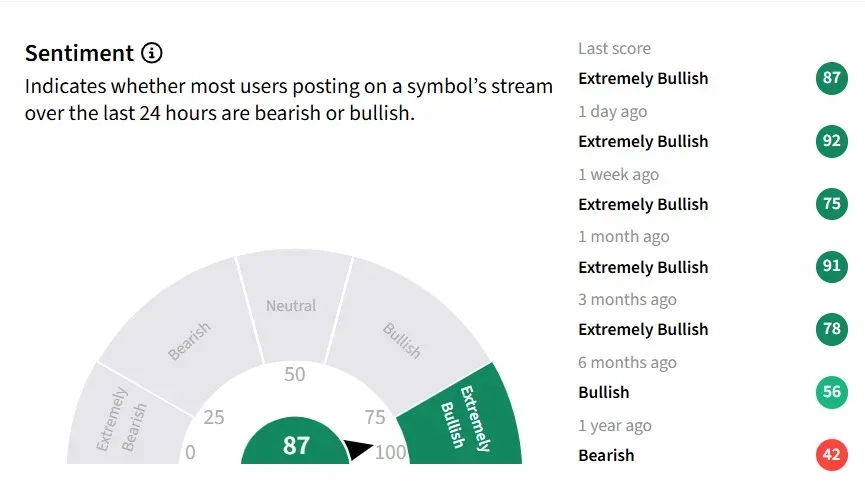
One user said the trial’s outcome is strong enough to justify considering Anktiva + BCG as the “standard of care in BCG-naive NMIBC.”
Advertisement|Remove ads.
Another user expects consolidation before the next leg of the rally.
Advertisement|Remove ads.
Year-to-date, the stock has shot up 350%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Comments posted here will also appear on symbol pages.

/filters:format(webp)https://news.stocktwits-cdn.com/large_Ark_Invest_871846bfd7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_michael_saylor_OG_4_jpg_292dd07afc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_grayscale_OG_jpg_c35dc5b1a2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_andre_francois_mckenzie_i_G_Yi_Bhd_N_Tp_E_unsplash_jpg_3d063f0b23.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1036973476_jpg_734afc6bfc.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Bitcoin_and_Ethereum_2b4356b70a.webp)