Advertisement|Remove ads.
Why Is PBM Stock More Than Doubling Premarket Today?

- Trump directed the FDA to expedite reviews of psychedelic medicines, including ibogaine, and committed $50 million in funding for clinical research.
- The company recently produced medical-grade ibogaine hydrochloride from an ethically sourced African supply.
- Last month, Psyence exported GMP-compliant psilocybin to Australia for use in a Phase IIb clinical trial.
Shares of Psyence Biomedical (PBM) surged over 111% to top premarket gains across U.S. stocks on Monday after U.S. President Donald Trump’s executive order accelerating psychedelic therapy research spotlighted the supply infrastructure behind post-traumatic stress disorder (PTSD) treatments such as ibogaine.
PBM stock surged 185% last week, marking its strongest weekly performance in more than two years.
Trump Commits $50M To Psychedelic Research
Over the weekend, Trump signed an executive order directing the U.S. Food and Drug Administration (FDA) to expedite the review of psychedelic medicines including ibogaine, while committing $50 million in federal funding toward related clinical research programs.
Speaking at the Oval Office signing ceremony, Trump said emerging psychedelic treatments could have a “tremendous impact” if supported by clinical evidence and highlighted their potential role in addressing serious mental-health conditions affecting veterans.
Ibogaine, derived from a shrub native to Central and West Africa, remains classified as a Schedule I substance in the U.S. despite growing interest from veterans’ advocacy groups studying its potential use in treating PTSD.
PBM Manufacturing Strategy In Spotlight
A day before the executive order was signed, Psyence CEO Jody Aufrichtig said, “Ibogaine has emerged as a compound of increasing global interest, particularly for its potential to address complex and treatment-resistant conditions.” He added that expanding regulated clinical research would be essential to improving both patient safety and long-term access to ibogaine-based treatments.
Through its investment in PsyLabs, the company has built a Good Manufacturing Practice (GMP)-compliant platform that produces pharma-grade psychedelic compounds, including ibogaine, for use in clinical research and drug-development programs.
In January, the company said it produced high-purity ibogaine hydrochloride, a medical-grade form of the compound, from ethically sourced natural supply in Africa. The company had already secured access to high-potency iboga bark and established ibogaine operations in Africa through PsyLabs.
Separately, last month, the company exported GMP-compliant psilocybin product NPX-5 to Australia for use in an ongoing Phase IIb clinical trial evaluating treatment for adjustment disorder in patients with cancer receiving palliative care.
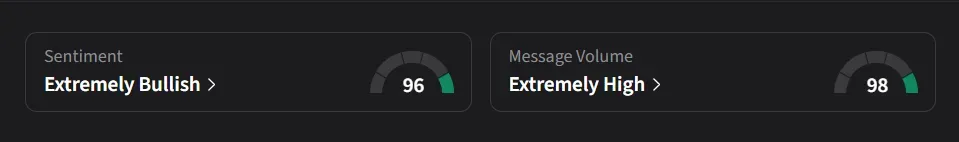
How Do Retail Traders Feel About PBM?
On Stocktwits, retail sentiment for PBM has been ‘extremely bullish’ for a week amid a whopping 15,100% surge in message volumes over the past month.
One user noted that, “psyense is emphasizing their fully integrated supply chain with GMP Able to produce ibogaine at commercial scale.”
Another user said, “Not only is it signed they’re bringing the drug to market in weeks NOT years!!! This is gonna be up 500% on Monday”
PBM stock has declined 65% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: DFTX Vs CMPS: Which Stock Has More Upside From Trump’s New Psychedelic Therapy Push?

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)